Summary
N-Acetylcysteine was given intravenously and as three fast dissolving and one slow-release formulation, on separate occasions, as a single dose of 600 mg to 10 fasting (5 men and 5 women) healthy volunteers. Blood and urine were sampled for the following 12 h.
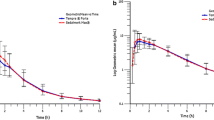
Renal clearance constituted around 30% of total body clearance, which was 0.21 l/h/kg. Volume of distribution was 0.33 l/kg, consistent with distribution mainly to extracellular water. The late elimination half-life was 2.27 h and the mean residence time 1.62 h.
The slow-release tablet resulted in a flattened plasma concentration-time curve typical of slow release formulations, while the other three oral formulations were rapidly absorbed.
The oral availability of N-acetylcysteine varied between 6 and 10%, with the slow-release tablet having the lowest and the fast dissolving tablet the highest availability.
Similar content being viewed by others
References
Sheffner AL (1983) The reduction in vitro in viscosity of mucoprotein solutions by a new mucolytic agent N-acetyl-L-cysteine. Ann NY Acad Sci 106: 298–310
Prescott LF, Illingworth RN, Critchley JAJH, Stewart MJ, Adam RD, Proudfoot AT (1979) Intravenous N-acetylcysteine: The treatment of choice for paracetamol poisoning. Br Med J 2: 1097–1100
Myers C, Bonow R, Palmeri S, Jenkins J, Corden B, Locker G, Doroshow J, Epstein S (1983) A randomized controlled trial assessing the prevention of doxorubicin cardiomyopathy by N-Acetylcysteine. Semin Oncol 10 [Suppl 1]: 53–55
Holoye PY, Duelge J, Hansen RM, Anderson T (1983) Prophylaxis, of ifosfamide toxicity with oral acetylcysteine. Semin Oncol 10 [Suppl 1]: 66–71
Botta JA, Nelson LW, Wieker JH Jr (1973) Acetylcysteine in the prevention of cyclophosphamide-induced cystitis in rats. J Natl Cancer Inst 51: 1051–1058
Grassi C, Morandi GC (1976) A controlled trial of intermittent oral acetylcysteine in the long-term treatment of chronic bronchitis. Eur J Clin Pharmacol 9: 393–396
Lemy-Debois N, Frigerio G, Lualdi P (1978) Oral acetylcysteine in bronchopulmonary disease. Comparative clinical trial with bromhexine. Acta Therapeut 4: 125–132
Multicenter Study Group (1980) Long-term oral acetylcysteine in chronic bronchitis. A double-blind controlled study. Eur J Respir Dis 61 [Suppl 111]: 93–108
Boman G, Bäcker U, Larsson S, Melander B, Wåhlinder L (1983) Oral acetylcysteine reduces exacerbation rates in chronic bronchitis: Report of a trial organized by the Swedish Society for Pulmonary Diseases. Eur J Respir Dis 64: 405–415
Rodenstein D, De Coster A, Gazzaniga A (1978) Pharmacokinetics of oral acetylcysteine: Absorption, binding and metabolism in patients with respiratory disorders. Clin Pharmacokinet 3: 247–254
Maddock J (1980) Biological properties of acetylcysteine: Assay development and pharmacokinetic studies. Eur J Respir Dis 61 [Suppl 111]: 52–58
Morgan LR, Holdiness MR, Gillen LE (1983) N-Acetylcysteine: Its bioavailability and interaction with ifosfamide metabolites. Semin Oncol 10 [Suppl 1]: 56–61
Frank H, Thiel D, Langer K (1984) Determination of N-acetyl-L-cysteine in biological fluids. J Chromatogr 309: 261–267
Kågedal B, Källberg M, Mårtensson J (1984) Determination of non-protein-bound N-acetylcysteine in plasma by high-performance liquid chromatography. J Chromatogr 311: 170–175
Lewis PA, Woodward AJ, Maddock J (1984) High-performance liquid chromatographic assay for N-acetylcysteine in plasma and urine. J Pharm Sci 73 [7]: 996–998
Borgström L, Johansson C-G, Larsson H, Lenander R (1981) Pharmacokinetics of bendroflumethiazide after low oral doses. J Pharmacokinet Biopharmaceut 9 [4]: 431–441
Kågedal B, Källberg M (1982) Reversed-phase ion-pair high-performance liquid chromatography of mercaptoacetate and N-acetylcysteine after derivatization with N-(1-pyrene)maleimide and N-(7-methylamino-4-methyl-3-coumarinyl)-maleimide. J Chromatogr 229: 409–415
Krenzelok EP, North DS, Peterson RG (1980) The effect of activated charcoal administration on N-acetylcysteine serum levels in human subjects. Vet Hum Toxicol 22 [Suppl 2]: 56–58
Rowland M, Tozer TN (1980) Clinical pharmacokinetics concepts and applications. Lea & Febiger, Philadelphia, p 50
Rowland M, Tozer TN (1980) Clinical pharmacokinetics concepts and applications. Lea & Febiger, Philadelphia, p 149
Bonanomi L, Gazzaniga A (1980) Toxicological, pharmacokinetic and metabolic studies on acetylcysteine. Eur J Respir Dis 61 [Suppl 111]: 45–51
Sheffner AL, Medler EM, Baily KR, Gallo DG, Mueller AJ, Sarett HP (1966) Metabolic studies with acetylcysteine. Biochem Pharmacol 15: 1523–1535
Lauterburg BH, Corcoran GB, Mitchell JR (1983) Mechanism of action of N-acetylcysteine in the protection against the hepatotoxicity of acetaminophen in rats in vivo. J Clin Invest 71: 980–991
Grassi C, Morandini GC, Frigerio G (1973) Clinical evaluation of systemic acetylcysteine by different routes of administration. Curr Ther Res 15: 165–179
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Borgström, L., Kågedal, B. & Paulsen, O. Pharmacokinetics of N-acetylcysteine in man. Eur J Clin Pharmacol 31, 217–222 (1986). https://doi.org/10.1007/BF00606662
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00606662