Abstract
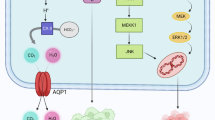
Prohibiting angiogenesis is an important therapeutic approach for fighting cancer and other angiogenic related diseases. Research focused on proteins that regulate abnormal angiogenesis has attracted intense interest in both academia and industry. Such proteins are able to target several angiogenic factors concurrently, thereby increasing the possibility of therapeutic success. Aquaporin-1 (AQP1) is a water channel membrane protein that promotes tumour angiogenesis by allowing faster endothelial cell migration. In this study we test the hypothesis that AQP1 inhibition impairs tumour growth in a mouse model of melanoma. After validating the inhibitor efficacy of two different AQP1 specific siRNAs in cell cultures, RNA interference experiments were performed by intratumoural injections of AQP1 siRNAs in mice. After 6 days of treatment, AQP1 siRNA treated tumours showed a 75 % reduction in volume when compared to controls. AQP1 protein level, in AQP1 knockdown tumours, was around 75 % that of the controls and was associated with a significant 40 % reduced expression of the endothelial marker, Factor VIII. Immunofluorescence analysis of AQP1 siRNA treated tumours showed a significantly lower microvessel density. Time course experiments demontrated that repeated injections of AQP1 siRNA over time are effective in sustaining the inhibition of tumour growth. Finally, we have confirmed the role of AQP1 in sustaining an active endothelium during angiogenesis and we have shown that AQP1 reduction causes an increase in VEGF levels. In conclusion, this study validates AQP1 as a pro-angiogenic protein, relevant for the therapy of cancer and other angiogenic-related diseases such as psoriasis, endometriosis, arthritis and atherosclerosis.
Similar content being viewed by others
Introduction
The speed of water transport across cell plasma membranes can be drastically increased by the expression of specific water channel proteins, called aquaporins (AQPs), allowing a bidirectional water flux only dependant on the osmotic and hydrostatic gradients [1]. AQPs have an obvious role in osmotically driven transepithelial fluid transport, such as in the kidney urinary concentrating mechanism and in glandular fluid secretion. Surprisingly, AQPs also play a role in several other cell mechanisms including brain swelling, neural signal transduction, skin moisturization and fat metabolism [2, 3]. Interestingly, a clear involvement of AQPs in tumour angiogenesis [4–7] as well as in cell migration has recently indicated these proteins as new players in tumour biology [8]. In particular, studies performed on AQP null mice and cell culture models have demonstrated that AQP overexpression facilitates cell migration which in turn plays a pivotal role in tumour spreading as well as in tumour angiogenesis [5]. The proposed mechanism is based on the concomitant polarized expression at the leading edge of the cell of AQPs and several transporters involved in migration, including Na+/H+ and Cl−/HCO3 −, supposed to enhance the water influx which is essential for the localized protrusions of plasma membranes. Furthermore, the ability to enhance cell motility is neither AQP nor cell-specific [5].
AQP altered expression has been associated to at least 12 different kinds of human tumours [8], with a mainly positive correlation between histological tumour grade and protein expression levels. The overexpression of AQPs has been revealed both at the level of the tumour cells forming the tumour mass itself and at the level of the proliferating endothelial cells forming the tumour vasculature. Altered AQP expression at the level of the tumour cells does not seem to be specific for a particular AQP. For instance, aberrant expression of AQP1 [9, 10], AQP4 [11], and AQP9 [12] have been found in human glioma. One hypothesis could be that AQP overexpression in tumour cells would allow water to rapidly penetrate into the growing tumour mass and cause tumour expansion. For example, in astrocytomas, AQP4 expression levels correlate with the extent of tumour associated brain edema but not with patient survival [11, 13]. In any case, the altered expression of AQPs in tumour cells remains a confused matter and no AQPs have as yet been validated as diagnostic or prognostic markers.
Tumour angiogenesis is one of the most important mechanisms for tumour development allowing tumour cells to receive the oxygen and nutrients necessary to survive and proliferate. Tumour endothelial cells arise from the normal endothelium of the surrounding host tissue thanks to cancerous cells secreting the molecular regulators of angiogenesis. This scenario is, however, complicated by alternative pathways, such as vasculogenic mimicry, which is the ability described for melanoma cells to mimic the activities of endothelial cells, therfore participating in neovascularization [14]. It seems that the greater the level of tumour angiogenesis in the primary tumour, the worse is the clinical outcome in several solid tumours, including melanomas, gliomas, breast, ovarian, lung, bladder and prostate cancers and also in hematological malignancies such as leukemias, myeloma and myelofibrosis [15].
The data showing that the endothelial AQP, AQP1, is involved in tumour angiogenesis are clearer. Our research group first proposed AQP1 as a promoter of tumour angiogenesis when AQP1 over-expression was found at the level of the endothelial plasma membrane of patients with active multiple myeloma [5, 7]. In the paper of Vacca et al., [16] however, the endothelial overexpression of AQP1 was only associated with increased angiogenesis with no direct proof of its role in this mechanism. Remarkably, greatly slowed tumour growth and improved survival, associated with impaired angiogenesis and cell migration, was later demonstrated in AQP1 null mice after subcutaneous and intracranial tumour cell implantation [5]. This strongly indicated, but once again did not directly demonstrate, that AQP1 inhibition could affect tumour angiogenesis and tumour growth in animals or humans affected by a cancer, still leaving this possibility to be proven with a direct approach.
In support of these findings, Heubert et al. [17] have recently shown that AQP1 upregulation facilitates pathological angiogenesis and invasion in cirrhotic liver by modulating the dynamics of FGF-dependent plasma membrane blebbing [17]. Interestingly, later studies, performed by the same authors on AQP1 null mice, have validated this protein as a logical treatment target in chronic liver disease and shown that the intense overexpression of AQP1 in cirrhotic endothelia depends on osmotically regulated microRNA expression [18].
We have previously shown that AQP1 knockdown (KD) induced impaired new vessel formation in the CAM (chick embryo chorioallantoic membrane), an in vivo model of physiological angiogenesis [19]. In the present study we present direct evidence that AQP1 inhibition impairs tumour growth in a mammalian tumour model where the tumour vasculature remains actively engaged in angiogenesis during melanoma cell growth [20]. By intratumoural injections of AQP1 specific siRNA in mice, we here demonstrate that constant AQP1 inhibition significantly interferes with tumour angiogenesis and tumour growth in vivo.
Materials and methods
Ethics statement
All experiments performed on animals were in line with the European Union Council Directive of 24 November 1986 (86/609/ EEC) on the ethical use of animals and were designed to minimise the number of animals used as well as their suffering. Experiments in this study were approved by the Italian Health Department (Art. 9 del Decreto Legislativo 116/92).
Cell lines
HeLa and B16F10 cells (both from ATCC, Rockville, MD, USA) were cultured in DMEM medium supplemented with 10 % fetal bovine serum (FBS), 100 U/ml penicillin and 100 mg/ml streptomycin, and maintained at 37 °C in a 5 % CO2 incubator. All the cell culture products were purchased from Invitrogen (Milan, Italy).
Short interfering RNAs
Two different AQP1 siRNA duplexes, specific for human and mouse AQP1 were purchased from Dharmacon Research, Inc. (Euroclone, Milan, Italy) and called AQP1 siRNA 1 (5’-GGGUGGAGAUGAAGCCCAAUU-3’) and AQP1 siRNA 2 (5’-UGGAGAUGAAGCCCAAAUAUU-3’). A scrambled siRNA (5’-UGGAGAAGGCCAACUAGGGUU-3’) was used as a negative control (CTRL siRNA). The selected siRNA sequences were submitted to a BLAST search to avoid the targeting of other homologous genes. A commercially available fluorescent siRNA was also used to follow its delivery into the tumour mass after intratumoural injection (BLOCK-iT Fluorescent Oligo, Invitrogen, Italy).
AQP1 transfection and RNA interference in HeLa cells
RNA interference (RNAi) experiments in HeLa cells were performed by transient co-transfection with mouse AQP1 cDNA subcloned into pTARGET expression vector (Promega, Milan, Italy) together with the siRNA sequence under analysis (100 nM). The Lipofectamine 2000 (Invitrogen, Milan, Italy) transfection protocol was used. Cells were analysed for AQP1 expression by Western blot analysis after 24, 48 and 72 hours.
Implantation of B16F10 tumour cells and RNAi experiments in mice
Experiments were performed on 8-week old male C57BL6/J mice purchased from Harlan (Calco, Italy). Mice were kept on a 12 h light–dark cycle with food and water ad libitum. 106 B16F10 cells in 200 μl PBS were injected subcutaneously into the back of the mice. Tumour length (L) and width (W) were measured using a caliper and the tumour volume was calculated as L x W2 × 0.5 [21].
RNAi experiments always started ten days after tumour cell implantation (day 0). 12 μg of CTRL siRNA or AQP1 si RNA (a 50 % mix of AQP1 siRNA 1 + 2) were complexed with 50 μl of Oligofectamine (Invitrogen, Milan, Italy) in a total volume of 200 μl of 5 % glucose in water. After 20 min of incubation, the siRNA-Oligofectamine complexes were injected in the tumour using an insulin syringe with a permanently attached needle. The time schedule of siRNA treatments was performed, depending on the analysis, as indicated in the figure legend. The following amount of siRNAs were also used: 12 μg on day 0 and day 3, 24 μg on day 6 and day 9, 48 μg on day 12. When increasing amounts of siRNA were used, the amount of Oligofectamine and the total volume were scaled up accordingly. For each experiment, 4 to 6 mice were dedicated to each condition (untreated, CTRL siRNA and AQP1 siRNA treated). For the analysis of siRNA delivery, the tumours were injected with 12 μg of the fluorescent siRNA and analyzed after 4 h by fluorescence microscopy.
Antibodies
The following primary antibodies were used: rabbit polyclonal anti-AQP1 (Santa Cruz, CA, USA), mouse monoclonal anti-Factor VIII (Millipore, Melbourne, Australia), rabbit polyclonal anti-actin (Sigma-Aldrich, Milan, Italy), goat polyclonal anti-VEGF (Santa Cruz, CA, USA). The following secondary antibodies were used: horseradish peroxidase (HRP) conjugated goat anti-rabbit and goat anti-mouse IgG (Santa Cruz Biotechnology, Santa Cruz, CA, USA), used for Western blot, and Alexa Fluor488 donkey anti-rabbit and Alexa Fluor594 donkey anti-mouse, used for Immunofluorescence (Invitrogen, Milan, Italy).
Proliferation assay
6 × 105 B16F10 cells were seeded in six-well plates (BD Falcon, Milan, Italy) and after 24 h transfected with 400 nM of siRNAs as previously described [22]. After 48 h the cells were trypsinized and subjected to cell counting using a Burker chamber.
Protein sample preparation
Cell cultures, extracted from tumours and normal skin were solubilized in at least seven volumes of RIPA buffer (25 mM Tris–HCl, pH 7.6; 150 mM NaCl; 1 % Triton X-100; 1 % sodium deoxycholate; 0.1 % SDS) added with a cocktail of protease inhibitors (Roche, Milan, Italy). The lysis was performed on ice for 1 h and the samples were then centrifuged at 22,000xg for 1 h. The protein content of the supernatant was measured with a bicinchoninic acid (BCA) Protein Assay Kit (Rockford, IL, USA).
Western blot analysis
Equal amounts of protein sample were separated by 12 % Tris-Glycine-SDS-PAGE and transferred to polyvinylidene difluoride membranes (Millipore, Milan, Italy) as described previously [23]. Membranes with blotted proteins were incubated with primary antibodies, washed, and incubated with peroxidase-conjugated secondary antibodies. Reactive proteins were revealed with an enhanced chemiluminescent detection system (ECL Plus; GE Healthcare, Buckinghamshire, UK) and visualized on a Versadoc imaging system (BioRad, Milan, Italy). Densitometric analysis was performed using Scion Image software (Frederick, MD, USA).
Immunofluorescence analysis
Tumours were fixed in 4 % paraformaldehyde and cryoprotected in 30 % sucrose. 8 μm transverse sections were prepared using a cryostat (CM 1900; Leica, Mannheim, Germany) and stored on positively charged glass slides (Thermo Scientific, Milan, Italy). Sections were treated as described [23]. Briefly, they were blocked using 0.1 % gelatin diluted in PBS for 30 min at RT, then incubated for 1 h with primary antibodies, washed with PBS-gelatin, incubated with Alexa Fluor488 and Alexa Fluor594 conjugated secondary antibodies and mounted with a medium containing 50 % Glycerol and 1 % n-propylgallate in PBS. Sections were examined with a Leica DMRXA photomicroscope equipped for epifluorescence and PL Fluotar 40 X and 100 X objectives (Leica, Germany). Digital images were obtained with a DX M1200 digital camera (Nikon, Japan) and ACT-1 version 2,20 software (Nikon, Japan). Once captured, the auto contrast function was applied to the whole images using Adobe Photoshop CS5. Sections containing the fluorescent siRNA were examined by confocal microscopy (TCS SP5, Leica, Germany).
Statistics
Data are expressed as mean ± SE. Statistically significant differences were computed using one-way Anova, the significance level being set at P < 0.05. The experiments were repeated from 3 to 5 times as indicated in the figure legend.
Results
Validation of two different AQP1 specific siRNAs in cell cultures
Before starting the AQP1 RNAi experiments on animals, we tested the efficacy of two different siRNA sequences in AQP1 expressing HeLa cells. Both sequences were designed to be specific for mouse and human AQP1. As shown in Fig. 1, the two AQP1 siRNAs either used individually or as a mixture of the two were able to reduce AQP1 protein expression to approximately 95 % of the control condition performed using a scrambled siRNA. The silencing was already high after 24 h and remained stable up to 72 h. This experiment shows that both the AQP1 siRNA sequences were able to strongly decrease AQP1 protein expression, with no significant differences between their use individually or as a 50 % mixture of the two.
AQP1 RNAi experiments in AQP1 expressing HeLa cells. (a) Immunoblot analysis of AQP1 expression in HeLa cells transiently transfected with two different siRNA sequences specific for AQP1, used individually (AQP1 siRNA1 or AQP1 siRNA 2) or as a 50 % mix (AQP1 siRNA 1 + 2). A scrambled siRNA (CTRL siRNA) was used as control. Actin was revealed in parallel for each sample and used as a non-targeted internal control housekeeping gene (not shown) for the densitometry analysis shown in B. (b) The histogram reports the densitometric analysis of the protein bands shown in A, calculated as the AQP1/Actin expression ratio. The histogram shows the analysis of 3–5 independent experiments performed 24, 48 and 72 h after transfection with the siRNAs (*P < 0.001 vs CTRL siRNA)
Impairment of tumour growth by intratumoural injection of AQP1 siRNAs in mice
For the in vivo experiments, we adopted the established tumour model in which B16F10 cells are injected into the shoulders of C57BL6/J mice. A visible tumour developed at the injection sites after 1 week, as expected. The first set of RNAi experiments was performed to verify the effective silencing of AQP1 at the tumour vasculature and the resulting effect on tumour angiogenesis and growth. The siRNAs were delivered by two intratumoural injections performed 10 (day 0) and 13 (day 3) days after the injection of the tumour cells. After three more days (day 6) the tumours were explanted and analyzed. As shown in Fig. 2a, the growth of the tumours treated with AQP1 siRNA (AQP1 siRNA 1 + AQP1 siRNA 2) was markedly affected compared to the CTRL siRNA treated or to untreated tumours.
Analysis of tumour growth in the murine model of melanoma. (a) Mice (top) and explanted tumours (bottom) after 6 days of treatment with siRNAs as indicated. The volume of the tumour is clearly reduced with AQP1 siRNA. (b) Diagram showing tumour growth over time of untreated (empty squares), CTRL siRNA treated (filled squares) and AQP1 siRNA treated (empty triangles) mice. The volume of the tumour was measured every day starting from the day of the first injection (day 0) until the last day before the explant (day 6). The arrows indicate the days (day 0 and day 3) when the siRNA injections were performed. (c) Histogram showing the tumour volume on day 6 (*P < 0.001, n = 4)
The analysis of the tumour volume over time (Fig. 2b) shows that the tumour growth was progressive and comparable between the untreated and CTRL siRNA treated tumours whereas it was significantly impaired in AQP1 siRNA treated ones. The last day before the explant and analysis, the volume of AQP1 siRNA treated tumours was one third that of untreated tumours or those treated with the CTRL siRNA (Fig. 2c). From qualitative analysis of the tumour shape we could not find any correlation between AQP1 expression and the shape of the tumour. We found CTRL siRNA, Untreated and AQP1 siRNA treated tumours of both nodular and spherical shapes most likely indicating a dependence on how the tumour cells established in the tissue at the moment of the s.c. injection.
Western blot experiments were performed to rule out a tumour angiogenesis independent effect due to direct action of AQP1 siRNA on B16F10 cells. Fig. 3a shows that AQP1 is not expressed in B16F10 cells and Fig. 3b reports a proliferation assay of the same cells performed in the presence of AQP1 siRNA. Both experiments indicate that the reduced tumour growth in mice was not due to a direct effect of AQP1 siRNA on the injected B16F10 tumour cells.
Analysis of the potential aspecific effect of AQP1 siRNA on B16F10 cells. (a) Western blot showing the absence of AQP1 expression in B16F10 cells. Mouse kidney was used as positive control for AQP1 expression. (b) Histogram summarizing a proliferation assay performed on B16F10 cells in the presence of AQP1 siRNA, CTRL siRNA and without siRNAs (Untreated). siRNAs do not affect B16F10 proliferation (P = 0.26, n = 3)
AQP1 KD impairs tumour vascularity in vivo
Tumours explanted on day six were analyzed to measure AQP1 expression at the level of tumour vessels and to investigate the resulting effect on tumour angiogenesis. AQP1 endothelial reduction was studied by Western blot and Immunofluorescence performed in parallel with the analysis of the endothelial marker, Factor VIII. Moreover, a fluorescent labelled siRNA was used to monitor siRNA delivery within the tumour.
The densitometric analysis of the Western blot results (Fig. 4a, b) showed that AQP1 protein expression levels were around 75 % lower in the tumours treated with AQP1 siRNA compared to the controls. AQP1 protein levels were similar in CTRL siRNA treated and untreated tumours, demonstrating the lack of toxic effect on the part of the lipid formulation and the RNA duplexes. Surprisingly, AQP1 siRNA treated tumours displayed a significant reduced expression of the endothelial marker Factor VIII, approximately 40 % that of the controls, indicating a reduced vascularity associated to AQP1 KD.
Analysis of AQP1 and Factor VIII expression and siRNA delivery within the tumour mass. (a) AQP1 and Factor VIII Western blot analysis of the tumours explanted on day 6 from mice treated as indicated in each lane. Actin was used as internal control for protein concentration. (b) The histogram summarizes the results of the densitometric analysis, of the bands revealed by Western blot reported as AQP1 or Factor VIII and actin ratio (*P < 0.05; **P < 0.005, n = 4). Note a significant reduction of Factor VIII associated with AQP1 KD. (c) AQP1 and Factor VIII double immunofluorescence experiments performed on cryosections of tumours explanted from mice treated as indicated. Also note no AQP1 expression outside the endothelial cells. The auto contrast function was only applied to the whole images using Adobe Photoshop CS5 without non-linear adjustments. (d) Histogram summarizing the number of vessels measured for each condition (*P < 0.001, n = 10). Scale bar: 200 μm. (e) Localization of fluorescent siRNA molecules (BLOCK-iT) injected in the tumour mass. Top and middle images: Confocal microscopy analysis showing that the siRNA fluorescence signal (green) is very intense at the level of the endothelial cells stained red with Factor VIII. Scale bar: 25 μm. Bottom image: the fluorescence signal is sparse and lower in the tumour cells analyzed by epifluorescence. Scale bar: 200 μm
AQP1 and Factor VIII double Immunofluorescence experiments were later performed to analyze the number of vessels of AQP1 siRNA treated tumours (Fig. 4c, d). The staining for AQP1 and Factor VIII in the control condition was very strong in all vessels localized primarily at the edges of the tumour, as expected. No AQP1 expression was found outside the endothelial cells within the tumour mass, indicating a targeted inhibition of endothelial AQP1. Interestingly, the tumours treated with AQP1 siRNA showed a significantly lower microvessel density associated to a heterogeneously altered dimension of the vessel diameter.
The use of a fluorescent siRNA (Fig. 4e) revealed a very efficient siRNA uptake by endothelial cells indicating that the diffusion of the lipid-siRNA formulation into the tumour is good enough to reach the periphery of the tumour even though it is injected into the centre of the tumour. The high endothelial siRNA uptake is consistent with the high level of endothelial AQP1 inhibition observed. As shown in the same image below, the tumour cells show a fluorescent signal which is lower and not homogeneous, indicating that such delivery of siRNA is not appropriate for targeting tumour cells.
All together these results indicate that the reduction of AQP1 protein induced by AQP1 siRNA significantly impairs tumour angiogenesis.
The inhibition of tumour growth can be prolonged over time by repeated injections of AQP1 siRNA
After assessing the efficacy of AQP1 inhibition in impairing tumour growth, we focused on the time course of tumour growth depending on the number of siRNA injections. The tumours were measured every three days starting from the first day of treatment with siRNAs (day 0) for approximately 15 days. We performed and compared two sets of experiments. In the first set only two injections were performed at the beginning of the tumour growth, day 0 and day 3. In the second set of experiments, the siRNA injections were performed every three days for the entire period under analysis. The results from the two sets of experiments are summarized in the diagram in Fig. 5a. When the treatment was limited to only two injections of AQP1 siRNA at day 0 and day 3, the tumours started growing again in a few days indicating that the siRNA effect is limited to 3–4 days and after that time is most likely degraded by the cells. In line with this indication, the parallel experiment performed by injecting progressively growing amounts of AQP1 siRNAs every three days for the entire period of observations demonstrated that the size of the tumour can be kept significantly lower compared to the control. To rule out the possibility that the size of the tumour was affected by hematoma caused by multiple injections, a control experiment was later performed in which the growth of CTRL siRNA tumours was compared with that of untreated ones. As shown in Fig. 5b, there is a complete overlapping of the size of the tumours from the two compared conditions at all days analyzed, indicating that the tumour size is not affected by injection provoked hematoma.
Analysis of tumour growth over time using two different experimental conditions for AQP1 siRNA injections. (a) Diagram showing tumour growth measured for 15 days after the first treatment with CTRL (square shapes) and AQP1 (round shapes) siRNAs. In the first set of experiments (empty shapes) only two injections were performed at the beginning of tumour growth (day 0 and day 3). Note in this case that the AQP1 siRNA treated tumours after the second treatment tended to reach the dimension of the control tumours. In the second set of experiments (filled shapes) the siRNA injections were performed every three days for the entire period of analysis. Note that in this case the size of the tumour was kept significantly lower than the controls. (b) The growth of uninjected (Untreated) and injected (CTRL siRNA) tumours was compared every three days during the fifteen days of analysis. A comparable growth between Untreated and CTRL siRNA treated tumours at all the stages analyzed, indicates that there is no hematoma dependent overgrowth due to repeated intratumoural injections
This experiment indicates that sustained AQP1 inhibition can certainly be effective in inhibiting tumour growth over time.
AQP1 is upregulated during tumour angiogenesis and its inhibition is directely responsible for reduced vascularity and increased VEGF levels
As an attempt to clarify the role of AQP1 in the B16F10 model during angiogenesis and to better understand the molecular events associated with AQP1 KD, we first investigated whether AQP1 expression levels were altered in tumour endothelial cells compared to the skin static endothelium. Western blot experiments showed strong AQP1 upregulation in tumours (Fig. 6a, b), in line with AQP1 endothelial upregulation already reported for multiple myeloma human biopsies [6], and in support for a primary role for AQP1 in sustaining an active endothelium during tumour angiogenesis. We later wanted to clarify the schedule of the events characterizing the reduction in vessel density observed after 6 days of RNAi shown in Fig 4c. To further confirm that the reduced vascularity is consequent to AQP1 inhibition and not vice versa, we analyzed tumours at day 2 of AQP1 siRNA treatment. The results confirm that at this stage, AQP1 started to reduce meanwhile the number of vessels was still normal (Fig. 6c), indicating that AQP1 KD is a forerunner to the reduced vascularity. Finally, western blot experiments were performed to assess whether a reduced expression of hypoxia responsive molecules such as VEGF could be correlated with the reduction in microvessels observed. As shown in Fig. 6d, e the expression level of VEGF was significantly upregulated in AQP1 siRNA treated tumours compared to the controls, most likely indicating a reaction of the tumour mass to the need for increasing the number of vessels.
AQP1 expression during tumour angiogenesis and analysis of the reduced vascularity and VEGF expression levels in AQP1 siRNA treated tumours (a) Representative Western blot showing AQP1 upregulated expression in melanoma compared to normal skin. (b) Histogram showing the densitometric analysis reported as the AQP1/Factor VIII ratio. Both AQP1 and Factor VIII were previously normalized on actin levels. The histogram shows the analysis of 3–5 independent experiments (*P < 0.05 vs normal skin). (c) Immunofluorescence experiments performed on tumours on day 2 of AQP1 siRNA treatment. The number of vessels does not show abnormalities at this stage of AQP1 inhibition while AQP1 appears reduced compared to the control conditions shown in Fig. 4c. (d) Western blot experiments showing the VEGF expression levels in CTRL siRNA and AQP1 siRNA treated tumours. (e) Histogram showing the densitometric analysis reported as VEGF/Actin levels (n = 6 , **P < 0.01 vs CTRL siRNA)
Discussion
In this study we provide direct proof that the inhibition of a water channel protein, AQP1, strongly impairs tumour growth by reducing the extent of neovascularization. We had already tested the effect of AQP1 inhibition in the chick embryo chorioallantoic membrane [19], a commonly used in vivo model of physiological angiogenesis, and found that AQP1 siRNA molecules were able to reduce angiogenesis in a condition very different from that shown in the present study where tumours are induced in vivo in mice and AQP1 inhibition is induced only after the developing of the tumour. Based on the technical approach here used and the results obtained, we believe that this study provides the first direct “proof of principle” that AQP1 inhibition impairs tumour growth as a cause-effect event. Starting from the technical approach used here, there are some aspects that need more detailed attention, for example the molecules to use for AQP1 inhibition and their delivery. We have used siRNA molecules complexed with lipid based formulation, delivered by intratumoural injections. Our choice to use siRNAs molecules, instead of plasmid or viral vectors expressing short hairpin RNA (shRNA), was driven by their much smaller size, which makes them easier to deliver and lacking the problems of insertional mutagenesis and immunogenicity that characterize viral vectors. On the other hand, we are also aware that the use of siRNAs, although considered promising due to their huge potential as therapeutic agents, is still in its early stages and therefore the discovery of chemical compounds or other molecules able to inhibit the function of AQP1 is of course envisaged. One of the problems related to the therapeutical use of siRNAs is their targeted delivery to specific organs. In this study our first choice was to deliver the siRNAs via intratumoural injections based on the easy accessibility of the tumour given by the mouse model, and also on our main purpose which was to demonstrate that AQP1 inhibition efficiently affects tumour development, without the intention of showing a therapeutic protocol. Additional information that we believe important to disclose is that AQP1 siRNA injections performed in the surrounding normal tissue and not in the tumour itself failed to knockdown AQP1 (data not shown), indicating that the diffusion of the lipid-siRNA formulation is most likely to be not as high in the normal tissue as it is in the tumour.
Obviously, for a wider range of applications, a systemic delivery of AQP1 inhibitors needs to be taken into account given that cancer is considered a systemic disease, including metastatic distribution of microdisseminated cells.
Thinking of a systemic and non-targeted delivery of antiangiogenic molecules, we believe AQP1 to be an alternative candidate to VEGF. Anti-VEGF therapies might interfere with the neurotrophic [24] and vasculotrophic [25] actions of VEGF and this needs attention, for example in patients at high risk of cardiac ischaemia or when a neuronal loss occurs [26]. In contrast, one could predict that anti-AQP1 therapies will not show particular adverse effect on the whole human organism, based on the analysis of AQP1 null humans (the so called “Colton null”) who do not display a clinical phenotype [27], even if, in a more detailed study, AQP1 null mice displayed a mild defect in the urinary concentrating mechanism [28]. Another important issue that makes AQP1 a good antiangiogenic candidate is related to its mechanism of action. The anti-VEGF therapy is based on the inhibition of the angiogenesis signaling cascade. However, as the clinical trials continue, it seems that many tumours are able to bypass therapeutic angiogeneses inhibition by activating or upregulating alternative proangiogenic pathways [29–31]. For example, reduced tumour angiogenesis has only been observed as an initial responce in mice treated with a monoclonal antibody able to block the VEGFR signaling cascade [32]. However, after a short period of time, the tumours show a reinitiation of tumour angiogeneses due to mRNA upregulation of the two proangiogenic factors FGF and Ang-1 [32]. For these reasons some patients become resistant to anti-VEGF treatment, therefore indicating the need to find VEGF-independent mechanisms leading to tumour angiogenesis.
The strong upregulation of AQP1 during tumour angiogenesis confirms its role in sustaining an active endothelium. The first mechanism of action explaining AQP1 implication in tumour angiogenesis might be linked to the migration of endothelial cells for the formation of new vessels [5]. The membrane protrusion, which is the first step for a cell to migrate, is facilitated by local osmotic gradients causing water entry. The presence or absence of AQP1 at the leading edge of the cell has an impact on the speed of water influx and therefore on local hydrostatic pressure which is essential for lamellipodium formation. Of course cell migration occurs even without AQPs but in this case the speed of the process is definitely slower. The second possibility to explain the role of AQP1 in tumour angiogenesis comes from the observed dramatic cytoskeleton alterations depending on AQP4 [22] and AQP1 [33] knockdown in cell cultures, and on studies indicating that F-actin cytoskeleton plays a primary role for AQP4 plasma membrane localization and during cell adhesion in astrocytes [34]. Based on these observations, it is the functional interaction of AQPs with cytoskeleton that is the key for the endothelial morphological changes which are a prerequisite for migration and neoangiogenesis to occur. Finally, the third mechanism of action explaining AQP1 implication in tumour angiogenesis is based on AQP1 upregulation found under hypoxic conditions [35], the explanation of which is based on the possibility that AQP1 could function as an O2 permeable channel [36]. The mechanism in this case would be that AQP1 upregulation would provide more oxygen under hypoxic conditions. Based on this relationship, we measured the level of hypoxia responsive molecules, such as VEGF [37], in AQP1 knockdown tumors. Interestingly, we found a significant increase in VEGF expression levels in AQP1 siRNA treated tumours. The mechanism we therefore propose, based on all the results here obtained is the following: 1) AQP1 plays a key role for an active endothelium; 2) AQP1 KD directly causes reduced vascularity; 3) the reduced vascularity is responsible for increased hypoxia, and 4) the increased hypoxia causes an increase in hypoxia responsive molecules, such as VEGF. The upregulation of VEGF depending on AQP1 KD is an important data to dissect the molecular pathway activated by AQP1 KD and also very important in view of the use of AQP1 as a target candidate for antiangiogenic therapy. It opens up the possibility that the inhibition of both AQP1 and VEGF at the same time could be a valid choice for optimal therapeutical results.
In conclusion, on the basis of the reported findings, we have validated AQP1 as a pro-angiogenic protein and therefore a possible target candidate for antiangiogenic molecules. Although we believe, in a more general way, that therapies addressing the multifactorial nature of tumour angiogenesis will be more effective than a single-molecule-specific method, these results provide a novel way to reduce tumour vessels and help the development of therapeutics for treating cancer and other angiogenic-related diseases such as psoriasis, endometriosis, arthritis, and atherosclerosis.
References
Amiry-Moghaddam M, Otsuka T, Hurn PD, Traystman RJ, Haug FM, Froehner SC, Adams ME, Neely JD, Agre P, Ottersen OP et al (2003) An alpha-syntrophin-dependent pool of AQP4 in astroglial end-feet confers bidirectional water flow between blood and brain. Proc Natl Acad Sci USA 100:2106–2111
Agre P, Kozono D (2003) Aquaporin water channels: molecular mechanisms for human diseases. FEBS Lett 555:72–78
Verkman AS (2005) More than just water channels: unexpected cellular roles of aquaporins. J Cell Sci 118:3225–3232
Endo M, Jain RK, Witwer B, Brown D (1999) Water channel (aquaporin 1) expression and distribution in mammary carcinomas and glioblastomas. Microvasc Res 58:89–98
Saadoun S, Papadopoulos MC, Hara-Chikuma M, Verkman AS (2005) Impairment of angiogenesis and cell migration by targeted aquaporin-1 gene disruption. Nature 434:786–792
Vacca A, Frigeri A, Ribatti D, Nicchia GP, Nico B, Ria R, Svelto M, Dammacco F (2001) Microvessel overexpression of aquaporin 1 parallels bone marrow angiogenesis in patients with active multiple myeloma. Br J Haematol 113:415–421
Vacca A, Ribatti D, Roccaro AM, Frigeri A, Dammacco F (2001) Bone marrow angiogenesis in patients with active multiple myeloma. Semin Oncol 28:543–550
Verkman AS, Hara-Chikuma M, Papadopoulos MC (2008) Aquaporins-new players in cancer biology. J Mol Med (Berl) 86:523–529
Oshio K, Binder DK, Liang Y, Bollen A, Feuerstein B, Berger MS, Manley GT (2005) Expression of the aquaporin-1 water channel in human glial tumours. Neurosurgery 56:375–381, discussion 375–381
Saadoun S, Papadopoulos MC, Davies DC, Bell BA, Krishna S (2002) Increased aquaporin 1 water channel expression in human brain tumours. Br J Cancer 87:621–623
Warth A, Simon P, Capper D, Goeppert B, Tabatabai G, Herzog H, Dietz K, Stubenvoll F, Ajaaj R, Becker R et al (2007) Expression pattern of the water channel aquaporin-4 in human gliomas is associated with blood–brain barrier disturbance but not with patient survival. J Neurosci Res 85:1336–1346
Warth A, Mittelbronn M, Hulper P, Erdlenbruch B, Wolburg H (2007) Expression of the water channel protein aquaporin-9 in malignant brain tumours. Appl Immunohistochem Mol Morphol 15:193–198
Saadoun S, Papadopoulos MC, Watanabe H, Yan D, Manley GT, Verkman AS (2005) Involvement of aquaporin-4 in astroglial cell migration and glial scar formation. J Cell Sci 118:5691–5698
Hendrix MJ, Seftor EA, Hess AR, Seftor RE (2003) Vasculogenic mimicry and tumour-cell plasticity: lessons from melanoma. Nat Rev Cancer 3:411–421
Kerbel RS (2000) Tumour angiogenesis: past, present and the near future. Carcinogenesis 21:505–515
Vacca A, Ribatti D, Roccaro AM, Ria R, Palermo L, Dammacco F (2001) Bone marrow angiogenesis and plasma cell angiogenic and invasive potential in patients with active multiple myeloma. Acta Haematol 106:162–169
Huebert RC, Vasdev MM, Shergill U, Das A, Huang BQ, Charlton MR, LaRusso NF, Shah VH (2010) Aquaporin-1 facilitates angiogenic invasion in the pathological neovasculature that accompanies cirrhosis. Hepatology 52:238–248
Huebert RC, Jagavelu K, Hendrickson HI, Vasdev MM, Arab JP, Splinter PL, Trussoni CE, Larusso NF, Shah VH (2011) Aquaporin-1 promotes angiogenesis, fibrosis, and portal hypertension through mechanisms dependent on osmotically sensitive microRNAs. Am J Pathol 179:1851–1860
Camerino GM, Nicchia GP, Dinardo MM, Ribatti D, Svelto M, Frigeri A (2006) In vivo silencing of aquaporin-1 by RNA interference inhibits angiogenesis in the chick embryo chorioallantoic membrane assay. Cell Mol Biol (Noisy-le-grand) 52:51–56
Langenkamp E, Vom Hagen FM, Zwiers PJ, Moorlag HE, Schouten JP, Hammes HP, Gouw AS, Molema G (2011) Tumour Vascular Morphology Undergoes Dramatic Changes during Outgrowth of B16 Melanoma While Proangiogenic Gene Expression Remains Unchanged. ISRN Oncol 2011:409308
Ikeda K, Nakano R, Uraoka M, Nakagawa Y, Koide M, Katsume A, Minamino K, Yamada E, Yamada H, Quertermous T et al (2009) Identification of ARIA regulating endothelial apoptosis and angiogenesis by modulating proteasomal degradation of cIAP-1 and cIAP-2. Proc Natl Acad Sci USA 106:8227–8232
Nicchia GP, Srinivas M, Li W, Brosnan CF, Frigeri A, Spray DC (2005) New possible roles for aquaporin-4 in astrocytes: cell cytoskeleton and functional relationship with connexin43. FASEB J 19
Nicchia GP, Mastrototaro M, Rossi A, Pisani F, Tortorella C, Ruggieri M, Lia A, Trojano M, Frigeri A, Svelto M (2009) Aquaporin-4 orthogonal arrays of particles are the target for neuromyelitis optica autoantibodies. Glia 57:1363–1373
Carmeliet P, Tessier-Lavigne M (2005) Common mechanisms of nerve and blood vessel wiring. Nature 436:193–200
Alon T, Hemo I, Itin A, Pe’er J, Stone J, Keshet E (1995) Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nat Med 1:1024–1028
Gariano RF, Gardner TW (2005) Retinal angiogenesis in development and disease. Nature 438:960–966
Agre P, Smith BL, Preston GM (1995) ABH and Colton blood group antigens on aquaporin-1, the human red cell water channel protein. Transfus Clin Biol 2:303–308
Chou CL, Knepper MA, Hoek AN, Brown D, Yang B, Ma T, Verkman AS (1999) Reduced water permeability and altered ultrastructure in thin descending limb of Henle in aquaporin-1 null mice. J Clin Invest 103:491–496
Bergers G, Hanahan D (2008) Modes of resistance to anti-angiogenic therapy. Nat Rev Cancer 8:592–603
Carmeliet P (2005) Angiogenesis in life, disease and medicine. Nature 438:932–936
Prager G.W. PM, Unseld M., Zielinski C.C. (2011) Angiogenesis in cancer: Anti-VEGF escape mechanisms. Transl Lung Cancer Res
Casanovas O, Hicklin DJ, Bergers G, Hanahan D (2005) Drug resistance by evasion of antiangiogenic targeting of VEGF signaling in late-stage pancreatic islet tumours. Cancer Cell 8:299–309
La Porta C (2010) AQP1 is not only a water channel: It contributes to cell migration through Lin7/beta-catenin. Cell Adh Migr 4:204–206
Nicchia GP, Rossi A, Mola MG, Procino G, Frigeri A, Svelto M (2008) Actin cytoskeleton remodeling governs aquaporin-4 localization in astrocytes. Glia 56:1755–1766
Abreu-Rodriguez I, Sanchez Silva R, Martins AP, Soveral G, Toledo-Aral JJ, Lopez-Barneo J, Echevarria M (2011) Functional and transcriptional induction of aquaporin-1 gene by hypoxia; analysis of promoter and role of Hif-1alpha. PLoS One 6:e28385
Wang Y, Cohen J, Boron WF, Schulten K, Tajkhorshid E (2007) Exploring gas permeability of cellular membranes and membrane channels with molecular dynamics. J Struct Biol 157:534–544
Semenza GL (2001) Regulation of hypoxia-induced angiogenesis: a chaperone escorts VEGF to the dance. J Clin Invest 108:39–40
Acknowledgments
This work was supported by grants from “Progetto di Ricerca IDEA Giovani Ricercatori (GRBA085SIS)”, “Rete Nazionale di Proteomica (RBRN07BMCT_009)”, “FIRB Idee Progettuali (RBIP0695BB_004)” and by the Apulia region grant “Progetto Strategico APQ Ricerca (Neurobiotech) [PS124]”. The authors would like to thank Richard Lusardi for his assistance in revising the English of the article and Gaetano De Vito for his excellent technical assistance.
Disclosure Statement
The authors declare they have no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nicchia, G.P., Stigliano, C., Sparaneo, A. et al. Inhibition of aquaporin-1 dependent angiogenesis impairs tumour growth in a mouse model of melanoma. J Mol Med 91, 613–623 (2013). https://doi.org/10.1007/s00109-012-0977-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-012-0977-x