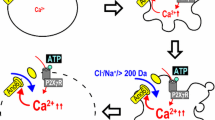
Abstract
The P2X7 receptor (P2X7R), an ATP-gated cation channel, is expressed predominantly in leukocytes. Activation of P2X7R has been implicated in the formation of a cytolytic pore (i.e., a large conductance channel) that allows the passage of molecules up to 900 Da in macrophages. At least two hypotheses have been presented to explain the conversion of a nonselective cation channel to a cytolytic pore. One hypothesis suggests that the pore is a separate molecular structure activated by P2X7R, and the second asserts that this is an intrinsic property of P2X7R (pore dilation). Based on connexin knockout and hemichannel antagonist studies, some groups have concluded that connexins and pannexins, the hemichannel-forming proteins in vertebrates, are fundamental components of the large conductance channel associated with P2X7R. Dye uptake and electrophysiology experiments were used to evaluate the efficacy and specificity of some hemichannel antagonists under conditions known to open the large conductance channel associated with P2X7R. Hemichannel antagonists and interference RNA (RNAi) targeting pannexin-1 did not affect P2X7R macroscopic currents [ATP, 1,570 ± 189 pA; ATP + 100 μM carbenoxolone (CBX), 1,498 ± 100 pA; ATP + 1 mM probenecid (Prob), 1,522 ± 9 pA] or dye uptake in a FACS assay (ATP, 63 ± 5 %; ATP + 100 μM CBX, 51.51 ± 8.4 %; ATP + 1 mM Prob, 57.7 ± 4.3 %) in mouse macrophages. These findings strongly suggest that the high-permeability pore evident after prolonged P2X7R activation does not occur through connexin or pannexin hemichannels in murine macrophages. Another membrane protein may be involved in P2X7R pore formation.






Similar content being viewed by others
Abbreviations
- P2X7R:
-
P2X7 receptor
- ox-ATP:
-
Adenosine 5′-triphosphate, periodate oxidized sodium salt
- CBX:
-
Carbenoxolone
- LY:
-
Lucifer yellow
- EB:
-
Ethidium bromide
- PI:
-
Propidium iodide
- 18α-GA:
-
18 α-Glycyrrhetinic acid
- BBG:
-
Brilliant Blue G
- Mef:
-
Mefloquine
- Prob:
-
Probenecide
- RNAi:
-
Interference RNA
- Mac:
-
Macrophage
- BSA:
-
Bovine serum albumin
- PBS:
-
Phosphate-buffered saline
- Panx1:
-
Pannexin-1
- Lipof:
-
Lipofectamine
References
Alloisio S et al (2008) Functional evidence for presynaptic P2X7 receptors in adult rat cerebrocortical nerve terminals. FEBS Lett 582(28):3948–3953
Alves LA et al (1995) Functional gap junctions in thymic epithelial cells are formed by connexin 43. Eur J Immunol 25(2):431–437
Alves LA et al (1996) Are there functional gap junctions or junctional hemichannels in macrophages? Blood 88(1):328–334
Alves LA, Coutinho-Silva R, Savino W (1999) Extracellular ATP: a further modulator in neuroendocrine control of the thymus. Neuroimmunomodulation 6(1–2):81–89
Atkinson L et al (2004) Differential co-localisation of the P2X7 receptor subunit with vesicular glutamate transporters VGLUT1 and VGLUT2 in rat CNS. Neuroscience 123(3):761–768
Auger R et al (2005) A role for mitogen-activated protein kinase(Erk1/2) activation and non-selective pore formation in P2X7 receptor-mediated thymocyte death. J Biol Chem 280(30):28142–28151
Barbe MT, Monyer H, Bruzzone R (2006) Cell-cell communication beyond connexins: the pannexin channels. Physiology (Bethesda) 21:103–114
Bauer R et al (2005) Intercellular communication: the Drosophila innexin multiprotein family of gap junction proteins. Chem Biol 12(5):515–526
Bennett MV et al (1991) Gap junctions: new tools, new answers, new questions. Neuron 6(3):305–320
Beyer EC, Steinberg TH (1991) Evidence that the gap junction protein connexin-43 is the ATP-induced pore of mouse macrophages. J Biol Chem 266(13):7971–7974
Bosco D, Haefliger J-A, Meda P (2011) Connexins: key mediators of endocrine function. Physiol Rev 91(4):1393–1445
Bruzzone R, Dermietzel R (2006) Structure and function of gap junctions in the developing brain. Cell Tissue Res 326(2):239–248
Bruzzone R et al (2003) Pannexins, a family of gap junction proteins expressed in brain. Proc Natl Acad Sci U S A 100(23):13644–13649
Bruzzone R et al (2005) Pharmacological properties of homomeric and heteromeric pannexin hemichannels expressed in Xenopus oocytes. J Neurochem 92(5):1033–1043
Cankurtaran-Sayar S, Sayar K, Ugur M (2009) P2X7 receptor activates multiple selective dye-permeation pathways in RAW 264.7 and human embryonic kidney 293 cells. Mol Pharmacol 76(6):1323–1332
Cario-Toumaniantz C et al (1998) Non-genomic inhibition of human P2X7 purinoceptor by 17beta-oestradiol. J Physiol 508(Pt 3):659–666
Cockcroft S, Gomperts BD (1980) The ATP4+ receptor of rat mast cells. Biochem J 188(3):789–798
Contreras JE et al (2002) Metabolic inhibition induces opening of unapposed connexin 43 gap junction hemichannels and reduces gap junctional communication in cortical astrocytes in culture. Proc Natl Acad Sci U S A 99(1):495–500
Coutinho-Silva R, Persechini PM (1997) P2Z purinoceptor-associated pores induced by extracellular ATP in macrophages and J774 cells. Am J Physiol 273(6 Pt 1):C1793–C1800
Deuchars SA et al (2001) Neuronal P2X7 receptors are targeted to presynaptic terminals in the central and peripheral nervous systems. J Neurosci: Offic J Soc Neurosci 21(18):7143–7152
Evans WH, Martin PE (2002) Lighting up gap junction channels in a flash. Bioessays 24(10):876–880
Faria RX, de Farias FP, Alves LA (2005) Are second messengers crucial for opening the pore associated with P2X7 receptor? Am J Physiol Cell Physiol 288(2):C260–C271
Faria RX et al (2009) Pharmacological properties of a pore induced by raising intracellular Ca2+. Am J Physiol Cell Physiol 297(1):C28–C42
Ferrari D et al (1997) Extracellular ATP triggers IL-1 beta release by activating the purinergic P2Z receptor of human macrophages. J Immunol 159(3):1451–1458
Gudipaty L et al (2003) Essential role for Ca2+ in regulation of IL-1beta secretion by P2X7 nucleotide receptor in monocytes, macrophages, and HEK-293 cells. Am J Physiol Cell Physiol 285:C286–C299
Harris AL (2001) Emerging issues of connexin channels: biophysics fills the gap. Q Rev Biophys 34(3):325–472
Heppel LA, Weisman GA, Friedberg I (1985) Permeabilization of transformed cells in culture by external ATP. J Membr Biol 86(3):189–196
Humphreys BD et al (2000) Stress-activated protein kinase/JNK activation and apoptotic induction by the macrophage P2X7 nucleotide receptor. J Biol Chem 275(35):26792–26798
Iglesias R et al (2008) P2X7 receptor-Pannexin1 complex: pharmacology and signaling. Am J Physiol Cell Physiol 295(3):C752–C760
Iglesias R et al (2009a) Pannexin 1: the molecular substrate of astrocyte "hemichannels". J Neurosci 29(21):7092–7097
Iglesias R, Spray DC, Scemes E (2009b) Mefloquine blockade of Pannexin1 currents: resolution of a conflict. Cell Commun Adhes 16(5–6):131–137
Jacobson GM et al (2010) Connexin36 knockout mice display increased sensitivity to pentylenetetrazol-induced seizure-like behaviors. Brain Res 1360:198–204
Jiang LH et al (2005) N-methyl-D-glucamine and propidium dyes utilize different permeation pathways at rat P2X(7) receptors. Am J Physiol Cell Physiol 289(5):C1295–C1302
Ke HZ et al (2003) Deletion of the P2X7 nucleotide receptor reveals its regulatory roles in bone formation and resorption. Mol Endocrinol (Baltimore, Md) 17(7):1356–1367
Kim SY et al (2007) ATP released from beta-amyloid-stimulated microglia induces reactive oxygen species production in an autocrine fashion. Exp Mol Med 39(6):820–827
Li Q et al (2003) Cell-specific behavior of P2X7 receptors in mouse parotid acinar and duct cells. J Biol Chem 278(48):47554–47561
Locovei S et al (2007) Pannexin1 is part of the pore forming unit of the P2X(7) receptor death complex. FEBS Lett 581(3):483–488
Ma W et al (2009) Pharmacological characterization of pannexin-1 currents expressed in mammalian cells. J Pharmacol Exp Ther 328(2):409–418
Ma W et al (2012) Pannexin 1 forms an anion-selective channel. Pflugers Arch 463(4):585–592
Marcoli M et al (2008) P2X7 pre-synaptic receptors in adult rat cerebrocortical nerve terminals: a role in ATP-induced glutamate release. J Neurochem 105(6):2330–2342
Monif M et al (2009) The P2X7 receptor drives microglial activation and proliferation: a trophic role for P2X7R pore. J Neurosci 29(12):3781–3791
Nagasawa K, Escartin C, Swanson RA (2009) Astrocyte cultures exhibit P2X7 receptor channel opening in the absence of exogenous ligands. Glia 57(6):622–633
Pelegrin P, Surprenant A (2006) Pannexin-1 mediates large pore formation and interleukin-1beta release by the ATP-gated P2X7 receptor. EMBO J 25(21):5071–5082
Pelegrin P, Surprenant A (2007) Pannexin-1 couples to maitotoxin- and nigericin-induced Interleukin-1beta release through a dye uptake-independent pathway. J Biol Chem 282:2386–2394
Persechini PM et al (1998) Extracellular ATP in the lymphohematopoietic system: P2Z purinoceptors off membrane permeabilization. Braz J Med Biol Res 31(1):25–34
Qu Y et al (2011) Pannexin-1 is required for ATP release during apoptosis but not for inflammasome activation. J Immunol 186(11):6553–6561
Rassendren F et al (1997a) Identification of amino acid residues contributing to the pore of a P2X receptor. EMBO J 16(12):3446–3454
Rassendren F et al (1997b) The permeabilizing ATP receptor, P2X7. Cloning and expression of a human cDNA. J Biol Chem 272(9):5482–5486
Riedel T et al (2007) Kinetics of P2X7 receptor-operated single channels currents. Biophys J 92(7):2377–2391
Schachter J et al (2008) ATP-induced P2X7-associated uptake of large molecules involves distinct mechanisms for cations and anions in macrophages. J Cell Sci 121(Pt 19):3261–3270
Silverman WR et al (2009) The pannexin 1 channel activates the inflammasome in neurons and astrocytes. J Biol Chem 284(27):18143–18151
Steinberg TH et al (1987) ATP4+ permeabilizes the plasma membrane of mouse macrophages to fluorescent dyes. J Biol Chem 262(18):8884–8888
Suadicani SO, Brosnan CF, Scemes E (2006) P2X7 receptors mediate ATP release and amplification of astrocytic intercellular Ca2+ signaling. J Neurosci 26(5):1378–1385
Surprenant A et al (1996) The cytolytic P2Z receptor for extracellular ATP identified as a P2X receptor (P2X7). Science 272(5262):735–738
Valiunas V (2002) Biophysical properties of connexin-45 gap junction hemichannels studied in vertebrate cells. J Gen Physiol 119(2):147–164
Valiunas V, Weingart R (2000) Electrical properties of gap junction hemichannels identified in transfected HeLa cells. Pflugers Arch 440(3):366–379
Valiunas V et al (1999) Electrophysiological properties of gap junction channels in hepatocytes isolated from connexin32-deficient and wild-type mice. Pflugers Arch 437(6):846–856
Virginio C et al (1999a) Kinetics of cell lysis, dye uptake and permeability changes in cells expressing the rat P2X7 receptor. J Physiol 519(2):335–346
Virginio C et al (1999b) Pore dilation of neuronal P2X receptor channels. Nat Neurosci 2(4):315–321
Yan Z et al (2008) The P2X7 receptor channel pore dilates under physiological ion conditions. J Gen Physiol 132(5):563–573
Acknowledgments
This work was supported by grants from IOC/FIOCRUZ, FAPERJ, and CNPq.
Conflict of interest
The authors state no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PPT 1150 kb)
Rights and permissions
About this article
Cite this article
Alberto, A.V.P., Faria, R.X., Couto, C.G.C. et al. Is pannexin the pore associated with the P2X7 receptor?. Naunyn-Schmiedeberg's Arch Pharmacol 386, 775–787 (2013). https://doi.org/10.1007/s00210-013-0868-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-013-0868-x