Abstract
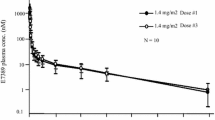
Dolastatins 10 and 15 are small peptides isolated from the marine sea hare Dolabella auricularia that have been shown to interact with tubulin. Their growth-inhibitory properties were compared using panels of human ovarian and colon-carcinoma cell lines. Both agents were very potent inhibitors of cell growth, with dolastatin 10 being an average of 9.1-fold more potent than dolastatin 15 [mean 50% inhibitory concentrations (IC50 values) 2.3×10–10 and 2.1×10–9 M, respectively; P <0.05] and more potent than paclitaxel or vinblastine. While neither dolastatin exhibited marked cross-resistance in cisplatin- or etoposide-resistant cell lines, contrasting effects were observed using an acquired doxorubicin-resistant (CH1doxR, 100-fold resistant, P-glycoprotein overexpressing) cell line. Resistance was significantly higher to dolastatin 15 (12.7-fold) than to dolastatin 10 (only 3.2-fold; P <0.05) and was reversible in both cases by verapamil. In vivo, using a s. c. advanced-stage human ovarian carcinoma xenograft and equitoxic doses, greater activity was observed with dolastatin 10 (6.1-day growth delay) versus 0.4 days for dolastatin 15. A radioimmunoassay for dolastatin 10 (limit of detection in mouse plasma 5 ng/ml) was developed. The rabbit antiserum also cross-reacted by 65% with dolastatin 15. Comparative mouse pharmacokinetics following i. v. administration of 1 mg/kg showed that both compounds are rapidly eliminated, but with a shorter second-phase half-life (t 1 /2 β) being observed for dolastatin 15 (being detectable for only up to 4 h post-administration), the t 1 /2 β being 3 times longer for dolastatin 10. In addition, areas under the plasma concentration-time curve (AUC values) were 1.6-fold higher for dolastatin 10 (333 versus 208 ng ml–1 h). Plasma binding of dolastatin 10 exceeded 90%. The highly sensitive RIA will be useful for pharmacokinetic studies in conjunction with the planned phase I clinical trials of these novel, extremely potent, tubulin-binding agents, of which dolastatin 10 appears to possess the more promising preclinical features.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 12 August 1995 / Accepted: 30 November 1995
Rights and permissions
About this article
Cite this article
Aherne, G., Hardcastle, A., Valenti, M. et al. Antitumour evaluation of dolastatins 10 and 15 and their measurement in plasma by radioimmunoassay. Cancer Chemother Pharmacol 38, 225–232 (1996). https://doi.org/10.1007/s002800050475
Issue Date:
DOI: https://doi.org/10.1007/s002800050475