Abstract
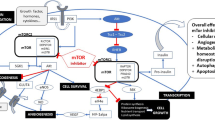
Mammalian target of rapamycin (mTOR) has emerged as an important target for cancer therapy. Rapamycin has a distinct, well-documented toxicity profile and most of the toxicity data has been reported in patients with organ transplantation. Newer mTOR inhibitors have slightly different pharmacokinetic properties, yet they present toxicity profiles similar to rapamycin. Most of these toxicities are mild to moderate in severity and can be managed clinically by dose modification and supportive measures. Mucositis and pneumonitis are the most commonly reported toxicities, but they rarely lead to treatment discontinuation. Pathogenesis of pneumonitis is uncertain, but various hypotheses have been suggested, including cell-mediated immune response to the drug.


Similar content being viewed by others
References
Hennessy BT et al (2005) Exploiting the PI3K/AKT pathway for cancer drug discovery. Nat Rev Drug Discov 4(12):988–1004
Scaltriti M, Baselga J (2006) The epidermal growth factor receptor pathway: a model for targeted therapy. Clin Cancer Res 12(18):5268–5272
Yu H, Rohan T (2000) Role of the insulin-like growth factor family in cancer development and progression. J Natl Cancer Inst 92(18):1472–1489
MacDonald AS (2001) A worldwide, phase III, randomized, controlled, safety and efficacy study of a sirolimus/cyclosporine regimen for prevention of acute rejection in recipients of primary mismatched renal allografts. Transplantation 71(2):271–280
Huang S, Houghton PJ (2002) Inhibitors of mammalian target of rapamycin as novel antitumor agents: from bench to clinic. Curr Opin Investig Drugs 3(2):295–304
Hudes G et al (2007) Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med 356(22):2271–2281
Raymond E et al (2004) Safety and pharmacokinetics of escalated doses of weekly intravenous infusion of CCI-779, a novel mTOR inhibitor, in patients with cancer. J Clin Oncol 22(12):2336–2347
Mita MM et al (2008) Phase I trial of the novel mammalian target of rapamycin inhibitor deforolimus (AP23573; MK-8669) administered intravenously daily for 5 days every 2 weeks to patients with advanced malignancies. J Clin Oncol 26(3):361–367
O'Donnell A et al (2008) Phase I pharmacokinetic and pharmacodynamic study of the oral mammalian target of rapamycin inhibitor everolimus in patients with advanced solid tumors. J Clin Oncol 26(10):1588–1595
Morelon E et al (2001) Characteristics of sirolimus-associated interstitial pneumonitis in renal transplant patients. Transplantation 72(5):787–790
Lennon A et al (2001) Interstitial pneumonitis associated with sirolimus (rapamycin) therapy after liver transplantation. Transplantation 72(6):1166–1167
Atkins MB, Stadler W et al (2002) A randomized double blind phase 2 study of intravenous CCI-779 administered weekly to patients with advanced renal cell carcinoma. Proc Am Soc Clin Oncol 21:36 Abstract
Atkins MB et al (2006) Randomized phase II study of multiple dose levels of CCI-779, a novel mammalian target of rapamycin kinase inhibitor, in patients with advanced refractory renal cell carcinoma. J Clin Oncol 22(5):909–918
Duran I et al (2004) Characterisation of the lung toxicity of the cell cycle inhibitor temsirolimus. Eur J Cancer 42(12):1875–1880
Pham PT et al (2004) Sirolimus-associated pulmonary toxicity. Transplantation 77(8):1215–1220
Motzer RJ et al (2008) Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. The Lancet 372(9637):449–456
Brattstrom C et al (1998) Hyperlipidemia in renal transplant recipients treated with sirolimus (rapamycin). Transplantation 65(9):1272–1274
Kraemer FB et al (1998) Insulin regulates lipoprotein lipase activity in rat adipose cells via wortmannin- and rapamycin-sensitive pathways. Metabolism 47(5):555–559
Massy ZA et al (2000) Hyperlipidaemia and post-heparin lipase activities in renal transplant recipients treated with sirolimus or cyclosporin A. Nephrol Dial Transplant 15(6):928
Hoogeveen RC et al (2001) Effect of sirolimus on the metabolism of apoB100- containing lipoproteins in renal transplant patients. Transplantation 72(7):1244–250
Kahan BD (2000) Efficacy of sirolimus compared with azathioprine for reduction of acute renal allograft rejection: a randomised multicentre study. The Rapamune US Study Group. Lancet 356(9225):194–202
Hidalgo M et al (2006) A phase I and pharmacokinetic study of temsirolimus (CCI-779) administered intravenously daily for 5 days every 2 weeks to patients with advanced cancer. Clin Cancer Res 12(19):5755–5763
23. Yee KW et al Phase I/II study of the mammalian target of rapamycin inhibitor everolimus (RAD001) in patients with relapsed or refractory hematologic malignancies. Clin Cancer Res 12(17):5165–5173
Fraenkel M et al (2008) mTOR inhibition by rapamycin prevents beta-cell adaptation to hyperglycemia and exacerbates the metabolic state in type 2 diabetes. Diabetes 57(4):945–957
Tremblay F, Marette A (2001) Amino acid and insulin signaling via the mTOR/p70 S6 kinase pathway. A negative feedback mechanism leading to insulin resistance in skeletal muscle cells. J Biol Chem 276(41):38052–38060
Galanis E et al (2005) Phase II trial of temsirolimus (CCI-779) in recurrent glioblastoma multiforme: a north central cancer treatment group study. J Clin Oncol 23(23):5294–5304
Bellmunt J et al (2008) Temsirolimus safety profile and management of toxic effects in patients with advanced renal cell carcinoma and poor prognostic features. Ann Oncol 19(8):1387–1392
Hong JC, Kahan BD (2000) Sirolimus-induced thrombocytopenia and leukopenia in renal transplant recipients: risk factors, incidence, progression, and management. Transplantation 69(10):2085–2090
Mita MM, Poplin E, Tap WD, Carmona A, Yonemoto L, Wages DS, Bedrosian CL, Rubin EH, Tolcher AW (2008) Deforolimus trial 106- A Phase I trial evaluating 7 regimens of oral Deforolimus (AP23573, MK-8669). J Clin Oncol 26:3509 abstract
Kneteman N, Babini R et al (2000) Sirolimus immunosuppression for liver transplantation in the presence of malignancy. XVIII International Congress of the Transplantation Society: Abstract
Stallone G et al (2005) Sirolimus for Kaposi's sarcoma in renal-transplant recipients. N Engl J Med 352(13):1317–1323
Stallone G et al (2008) Kaposi's sarcoma and mTOR: a crossroad between viral infection neoangiogenesis and immunosuppression. Transpl Int 21(9):825–832
Kahan BD, Camardo JS (2001) Rapamycin: clinical results and future opportunities. Transplantation 72(7):1181–1193
Groth CG et al (1999) Sirolimus (rapamycin)-based therapy in human renal transplantation: similar efficacy and different toxicity compared with cyclosporine. Sirolimus European renal transplant study group. Transplantation 67(7):1036–1042
Kreis H et al (2000) Sirolimus in association with mycophenolate mofetil induction for the prevention of acute graft rejection in renal allograft recipients. Transplantation 69(7):1252–1260
Morales JM, Wramner H, Kreis D et al (2000) Sirolimus vs cyclosporin: a comparision of renal function over two years. XVIII International Congress of the Transplantation Society 140:0428 Abstract
Chan S et al (2005) Phase II study of temsirolimus (CCI-779), a novel inhibitor of mTOR, in heavily pretreated patients with locally advanced or metastatic breast cancer. J Clin Oncol 23(23):5314–5322
Witzig TE et al (2005) Phase II trial of single-agent temsirolimus (CCI-779) for relapsed mantle cell lymphoma. J Clin Oncol 23(23):5347–5356
Amato RJ et al (2006) A phase II trial of RAD001 in patients (Pts) with metastatic renal cell carcinoma (MRCC). J Clin Oncol (Meeting Abstracts) 24((18_suppl)):4530
Chawla SP, Staddon AP, Schuetze SM, D'Amato GZ, Blay JY, Sankhala KK, Daly ST, Rivera VM, Demetri GD (2006) Updated results of a phase II trial of AP23573, a novel mTOR inhibitor, in patients (pts) with advanced soft tissue or bone sarcomas. J Clin Oncol 24(18S):9505
Rizzieri DA et al (2008) A phase 2 clinical trial of deforolimus (AP23573, MK-8669), a novel mammalian target of rapamycin inhibitor, in patients with relapsed or refractory hematologic malignancies. Clin Cancer Res 14(9):2756–2762
Conflict of interest statement
No funds were received in support of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sankhala, K., Mita, A., Kelly, K. et al. The emerging safety profile of mTOR inhibitors, a novel class of anticancer agents. Targ Oncol 4, 135–142 (2009). https://doi.org/10.1007/s11523-009-0107-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-009-0107-z