Abstract
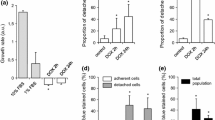
The cyclooxygenase (COX)-2 inhibitors celecoxib and rofecoxib were studied for their effects on neonatal rat cardiac myocytes as a possible model for the adverse cardiovascular effects that this class of compounds have shown in their clinical use. Celecoxib, but not rofecoxib, as measured by lactate dehydrogenase release was toxic to myocytes in the low micromolar concentration range. This toxicity shown by celecoxib was also associated with a high degree of myofibrillar disruption similar to that caused by doxorubicin. As measured by induction of caspase-3/7 activity and by changes in nuclear morphology, neither celecoxib nor rofecoxib strongly induced apoptosis in myocytes. The stable prostacyclin analog iloprost was unable to reduce celecoxib-induced damage, which suggested that celecoxib exerted its cytotoxicity through prostacyclin-independent pathways. Celecoxib treatment did not increase intracellular oxidation of 2′,7′-dichlorofluorescin in myocytes, which suggested that its cytotoxicity was not due to reactive oxygen species generation. The evidence supports the conclusion that celecoxib exerts its cytotoxicity towards myocytes through COX-2-independent mediated pathways.






Similar content being viewed by others
References
Adderley, S. R., & Fitzgerald, D. J. (1999). Oxidative damage of cardiomyocytes is limited by extracellular regulated kinases 1/2-mediated induction of cyclooxygenase-2. The Journal of Biological Chemistry, 274, 5038–5046.
Barnabé, N., Butler, M., & Hasinoff, B. B. (2001). The effect of the catalytic topoisomerase II inhibitor dexrazoxane (ICRF-187) on CC9C10 hybridoma viability and productivity. Cytotechnology, 37, 107–117.
Billingham, M., & Bristow, M. (1984). Evaluation of anthracycline cardiotoxicity: Predictive ability and functional correlation of endomycardial biopsy. Cancer Treatment Symposia, 3, 71–76.
Brooks, G., Poolman, R. A., & Li, J. M. (1998). Arresting developments in the cardiac myocyte cell cycle: Role of cyclin-dependent kinase inhibitors. Cardiovascular Research, 39, 301–311.
Chan, C. C., Boyce, S., Brideau, C., Charleson, S., Cromlish, W., Ethier, D., Evans, J., Ford-Hutchinson, A. W., Forrest, M. J., Gauthier, J. Y., Gordon, R., Gresser, M., Guay, J., Kargman, S., Kennedy, B., Leblanc, Y., Leger, S., Mancini, J., O’Neill, G. P., Ouellet, M., Patrick, D., Percival, M. D., Perrier, H., Prasit, P., Rodger, I., et al. (1999). Rofecoxib [Vioxx, MK-0966; 4-(4′-methylsulfonylphenyl)-3-phenyl-2-(5H)-furanone]: A potent and orally active cyclooxygenase-2 inhibitor. Pharmacological and biochemical profiles. The Journal of Pharmacology and Experimental Therapeutics, 290, 551–560.
Cocco, T., Di Paola, M., Papa, S., & Lorusso, M. (1999). Arachidonic acid interaction with the mitochondrial electron transport chain promotes reactive oxygen species generation. Free Radical Biology and Medicine, 27, 51–59.
Davies, N. M., McLachlan, A. J., Day, R. O., & Williams, K. M. (2000). Clinical pharmacokinetics and pharmacodynamics of celecoxib: A selective cyclo-oxygenase-2 inhibitor. Clinical Pharmacokinetics, 38, 225–242.
Davies, N. M., Teng, X. W., & Skjodt, N. M. (2003). Pharmacokinetics of rofecoxib: A specific cyclo-oxygenase-2 inhibitor. Clinical Pharmacokinetics, 42, 545–556.
Dowd, N. P., Scully, M., Adderley, S. R., Cunningham, A. J., & Fitzgerald, D. J. (2001). Inhibition of cyclooxygenase-2 aggravates doxorubicin-mediated cardiac injury in vivo. The Journal of Clinical Investigation, 108, 585–590.
Fosslien, E. (2005). Cardiovascular complications of non-steroidal anti-inflammatory drugs. Annals of Clinical and Laboratory Science, 35, 347–385.
Gabizon, A. A., Lyass, O., Berry, G. J., & Wildgust, M. (2004). Cardiac safety of pegylated liposomal doxorubicin (Doxil/Caelyx) demonstrated by endomyocardial biopsy in patients with advanced malignancies. Cancer Investigation, 22, 663–669.
Grosser, T., Fries, S., & FitzGerald, G. A. (2006). Biological basis for the cardiovascular consequences of COX-2 inhibition: therapeutic challenges and opportunities. The Journal of Clinical Investigation, 116, 4–15.
Hasinoff, B. B., Abram, M. E., Barnabé, N., Khelifa, T., Allan, W. P., & Yalowich, J. C. (2001). The catalytic DNA topoisomerase II inhibitor dexrazoxane (ICRF-187) induces differentiation and apoptosis in human leukemia K562 cells. Molecular Pharmacology, 59, 453–461.
Hasinoff, B. B., Abram, M. E., Chee, G.-L., Huebner, E., Byard, E. H., Barnabé, N., Ferrans, V. J., Yu, Z.-X., & Yalowich, J. C. (2000). The catalytic DNA topoisomerase II inhibitor dexrazoxane (ICRF-187) induces endopolyploidy in Chinese hamster ovary cells. The Journal of Pharmacology and Experimental Therapeutics, 295, 474–483.
Hasinoff, B. B., Patel, D., & Wu, X. (2003). The oral iron chelator ICL670A (deferasirox) does not protect myocytes against doxorubicin. Free Radical Biology and Medicine, 35, 1469–1479.
Kearney, P. M., Baigent, C., Godwin, J., Halls, H., Emberson, J.R., & Patrono, C. (2006). Do selective cyclo-oxygenase-2 inhibitors and traditional non-steroidal anti-inflammatory drugs increase the risk of atherothrombosis? Meta-analysis of randomised trials. British Medical Journal, 332, 1302–1308.
Knudsen, J. F., Carlsson, U., Hammarstrom, P., Sokol, G. H., & Cantilena, L. R. (2004). The cyclooxygenase-2 inhibitor celecoxib is a potent inhibitor of human carbonic anhydrase II. Inflammation, 28, 285–290.
Li, F., Wang, X., Capasso, J. M., & Gerdes, A. M. (1996). Rapid transition of cardiac myocytes from hyperplasia to hypertrophy during postnatal development. Journal of Molecular and Cell Cardiology, 28, 1737–1746.
Lim, C. C., Zuppinger, C., Guo, X., Kuster, G. M., Helmes, M., Eppenberger, H. M., Suter, T. M., Liao, R., & Sawyer, D. B. (2004). Anthracyclines induce calpain-dependent titin proteolysis and necrosis in cardiomyocytes. The Journal of Biological Chemistry, 279, 8290–8299.
Lin, H. P., Kulp, S. K., Tseng, P. H., Yang, Y. T., Yang, C. C., & Chen, C. S. (2004). Growth inhibitory effects of celecoxib in human umbilical vein endothelial cells are mediated through G1 arrest via multiple signaling mechanisms. Molecular Cancer Therapeutics, 3, 1671–1680.
Maier, T. J., Schilling, K., Schmidt, R., Geisslinger, G., & Grosch, S. (2004). Cyclooxygenase-2 (COX-2)-dependent and -independent anticarcinogenic effects of celecoxib in human colon carcinoma cells. Biochemical Pharmacology, 67, 1469–1478.
Mitchell, J. A., & Warner, T. D. (2006). COX isoforms in the cardiovascular system: Understanding the activities of non-steroidal anti-inflammatory drugs. Nature Reviews Drug Discovery, 5, 75–86.
O’Connell, T. D., Berry, J. E., Jarvis, A. K., Somerman, M. J., & Simpson, R. U. (1997). 1,25-Dihydroxyvitamin D3 regulation of cardiac myocyte proliferation and hypertrophy. The American Journal of Physiology, 272, H1751–H1758.
O’Malley, Y. Q., Reszka, K. J., & Britigan, B. E. (2004). Direct oxidation of 2′,7′-dichlorodihydrofluorescein by pyocyanin and other redox-active compounds independent of reactive oxygen species production. Free Radical Biology and Medicine, 36, 90–100.
Solomon, S. D., McMurray, J. J., Pfeffer, M. A., Wittes, J., Fowler, R., Finn, P., Anderson, W.F., Zauber, A., Hawk, E., & Bertagnolli, M. (2005). Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. The New England Journal of Medicine, 352, 1071–1080.
Subhashini, J., Mahipal, S. V., & Reddanna, P. (2005). Anti-proliferative and apoptotic effects of celecoxib on human chronic myeloid leukemia in vitro. Cancer Letters, 224, 31–43.
Tong, Z., Wu, X., Chen, C. S., & Kehrer, J. P. (2006). Cytotoxicity of a non-cyclooxygenase-2 inhibitory derivative of celecoxib in non-small-cell lung cancer A549 cells. Lung Cancer, 52, 117–124.
Wang, D., Wang, M., Cheng, Y., & Fitzgerald, G. A. (2005). Cardiovascular hazard and non-steroidal anti-inflammatory drugs. Current Opinion in Pharmacology, 5, 204–210.
Zhang, G. S., Liu, D. S., Dai, C. W., & Li, R. J. (2006). Antitumor effects of celecoxib on K562 leukemia cells are mediated by cell-cycle arrest, caspase-3 activation, and downregulation of Cox-2 expression and are synergistic with hydroxyurea or imatinib. American Journal of Hematology, 81, 242–255.
Acknowledgements
This work was supported by the Canadian Institutes of Health Research, the Canada Research Chairs program, the Province of Manitoba, through the Manitoba Research and Innovation Fund and a Canada Research Chair in Drug Development for Brian Hasinoff.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hasinoff, B.B., Patel, D. & Wu, X. The cytotoxicity of celecoxib towards cardiac myocytes is cyclooxygenase-2 independent. Cardiovasc Toxicol 7, 19–27 (2007). https://doi.org/10.1007/s12012-007-0002-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-007-0002-8