Abstract
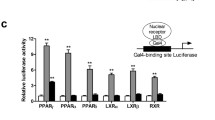
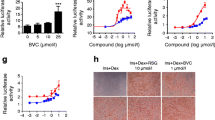
Peroxisome proliferator-activated receptor (PPAR) γ is known to be a key regulator of insulin resistance. PAR-1622 is a novel small molecule compound synthesized in Dong-A research center. In this study, we characterized the pharmacological profiles of PAR-1622, a selective partial activator of PPARγ. In transient transactivation assays, PAR-1622 [(S)-2-ethoxy-3(4-(5-(4-(5-(methoxymethyl)isoxazol-3-yl)phenyl)-3-methylthiophen-2-yl)methoxy)phenyl)propanoic acid] showed a partial activator against human PPARγ with an EC50 of 41 nM and a maximal response of 37% relative to the full agonist rosiglitazone without activating human PPARδ. PAR-1622 was 56 folds more selective for human PPARγ than for human PPARα (EC50, 2304 nM), which means that it is a selective partial activator of PPARγ. PAR-1622 also showed a partial activator against mouse PPARγ with an EC50 of 427 nM and a maximal response was 57% of that of rosiglitazone. INT-131, a selective PPARγ partial agonist in clinical stage, also was a partial activator against human PPARγ with an EC50 of 83 nM and a maximal response achieved by INT-131 was 49% of that observed with full agonist rosiglitazone. In functional assays using human mesenchymal stem cells, PAR-1622 induced adipocyte differentiation, which was 3-fold more potent with a comparable maximum response compared to INT-131. Furthermore, PAR-1622 significantly improved hyperglycemia in db/db when orally administered at a dose of 1 mg/kg/day for 5 days. In hemodilution assays with Evans Blue, rosiglitazone significantly increased the plasma volume in ICR mice that were orally administered 30 mg/kg/day for 9 days; however, PAR-1622 showed no significant effects on plasma volume, similar to INT-131. These results suggest that PAR-1622 is a selective partial activator of PPARγ and has excellent antihyperglycemic activities and a broad safety profile for fluid retention.
Similar content being viewed by others
References
Acton, J. J., 3rd, Black, R. M., Jones, A. B., Moller, D. E., Colwell, L., Doebber, T. W., Macnaul, K. L., Berger, J., and Wood, H. B., Benzoyl 2-methyl indoles as selective PPARgamma modulators. Bioorg. Med. Chem. Lett., 15, 357–362 (2005).
Allen, T., Zhang, F., Moodie, S. A., Clemens, L. E., Smith, A., Gregoire, F., Bell, A., Muscat, G. E., and Gustafson, T. A., Halofenate is a selective peroxisome proliferator-activated receptor gamma modulator with antidiabetic activity. Diabetes., 55, 2523–2533 (2006).
Bajaj, M., Suraamornkul, S., Hardies, L. J., Glass, L., Musi, N., DeFronzo, R. A., Effects of peroxisome proliferatoractivated receptor (PPAR)-alpha and PPAR-gamma agonists on glucose and lipid metabolism in patients with type 2 diabetes mellitus. Diabetologia., 50, 1723–1731 (2007).
Chen, L., Yang, B., McNulty, J. A., Clifton, L. G., Binz, J. G., Grimes, A. M., Strum, J. C., Harrington, W. W., Chen, Z., Balon, T. W., Stimpson, S. A., Brown, K. K., GI262570, a peroxisome proliferator-activated receptor {gamma} agonist, changes electrolytes and water reabsorption from the distal nephron in rats. J. Pharmacol. Exp. Ther., 312, 718–725 (2005).
Hung, Y. J., Lin, S. H., Pei, D., Kuo, S. W., Hsieh, C. H., He, C. T., Hsing Lee, C., Fan, S. C., and Sheu, W. H., Rosiglitazone improves insulin sensitivity in nonobese subjects with impaired glucose tolerance: the role of adiponectin and C-reactive protein. Metabolism., 55, 439–444 (2006).
Iwasaki, K., Zhang, R., Perhonen, M. A., Zuckerman, J. H., Levine, B. D., Reduced baroreflex control of heart period after bed rest is normalized by acute plasma volume restoration. Am J. Physiol. Regul. Integr. Comp. Physiol., 287, R1256–1262 (2004).
Knouff, C. and Auwerx, J., Peroxisome proliferatoractivated receptor-gamma calls for activation in moderation: lessons from genetics and pharmacology. Endocr. Rev., 25, 899–918 (2004).
Li, Y., Wang, Z., Motani, A., McGee, L. R., Learned, M., Timmermans, P. B., and Chen, J. L., T0903131 (T131): A selective modulator of PPARgamma. 64th Annu. Meet Sci. Sess. Am. Diabetes Assoc., Abst 659-P (2004).
Liu, K., Black, R. M., Acton, J. J., 3rd, Mosley, R., Debenham, S., Abola, R., Yang, M., Tschirret-Guth, R., Colwell, L., Liu, C., Wu, M., Wang, C. F., MacNaul, K. L., McCann, M. E., Moller, D. E., Berger, J. P., Meinke, P. T., Jones, A. B., and Wood, H. B., Selective PPARgamma modulators with improved pharmacological profiles. Bioorg. Med. Chem. Lett., 15, 2437–2440 (2005).
Merrill, D. C. and Cowley, A. W., Jr., Chronic effects of vasopressin on fluid volume distribution in conscious dogs. Am. J. Physiol., 252, F26–31 (1987).
Misra, P., Chakrabarti, R., Vikramadithyan, R. K., Bolusu, G., Juluri, S., Hiriyan, J., Gershome, C., Rajjak, A., Kashireddy, P., Yu, S., Surapureddi, S., Qi, C., Zhu, Y. J., Rao, M. S., Reddy, J. K., and Ramanujam, R., PAT5A: a partial agonist of peroxisome proliferator-activated receptor gamma is a potent antidiabetic thiazolidinedione yet weakly adipogenic. J. Pharmacol. Exp. Ther., 306, 763–771 (2003).
Mittra, S., Sangle, G., Tandon, R., Sharma, S., Roy, S., Khanna, V., Gupta, A., Sattigeri, J., Sharma, L., Priyadarsiny, P., Khattar, S. K., Bora, R. S., Saini, K. S., and Bansal, V. S., Increase in weight induced by muraglitazar, a dual PPARalpha/gamma agonist, in db/db mice: adipogenesis/or oedema? Br. J. Pharmacol., 150, 480–487 (2007).
Nesto, R. W., Bell, D., Bonow, R. O., Fonseca, V., Grundy, S. M., Horton, E. S., Le Winter, M., Porte, D., Semenkovich, C. F., Smith, S., Young, L. H., and Kahn, R., Thiazolidinedione use, fluid retention, and congestive heart failure: a consensus statement from the American Heart Association and American Diabetes Association. October 7, 2003. Circulation., 108, 2941–2948 (2003).
Nissen, S. E., Wolski, K., and Topol, E. J., Effect of muraglitazar on death and major adverse cardiovascular events in patients with type 2 diabetes mellitus. JAMA., 294, 2581–2586 (2005).
Olefsky, J. M., Treatment of insulin resistance with peroxisome proliferator-activated receptor gamma agonists. J. Clin. Invest., 106, 467–472 (2000).
Porte, D., Jr. and Schwartz, M. W., Diabetes complications: why is glucose potentially toxic? Science., 272, 699–700 (1996).
Rangwala, S. M. and Lazar, M. A., Peroxisome proliferatoractivated receptor gamma in diabetes and metabolism. Trends Pharmacol. Sci., 25, 331–336 (2004).
Rosenson, R. S., Otvos, J. D., and Freedman, D. S., Relations of lipoprotein subclass levels and low-density lipoprotein size to progression of coronary artery disease in the Pravastatin Limitation of Atherosclerosis in the Coronary Arteries (PLAC-I) trial. Am. J. Cardiol., 90, 89–94 (2002).
Tannehill-Gregg, S. H., Sanderson, T. P., Minnema, D., Voelker, R., Ulland, B., Cohen, S. M., Arnold, L. L., Schilling, B. E., Waites, C. R., and Dominick, M. A., Rodent carcinogenicity profile of the antidiabetic dual PPAR alpha and gamma agonist muraglitazar. Toxicol. Sci., 98, 258–270 (2007).
Tontonoz, P., Hu, E., Graves, R. A., Budavari, A. I., and Spiegelman, B. M., mPPAR gamma 2: tissue-specific regulator of an adipocyte enhancer. Genes Dev., 8, 1224–1234 (1994).
Xu, H. E., Lambert, M. H., Montana, V. G., Plunket, K. D., Moore, L. B., Collins, J. L., Oplinger, J. A., Kliewer, S. A., Gampe, R. T. Jr., McKee, D. D., Moore, J. T., and Willson, T. M., Structural determinants of ligand binding selectivity between the peroxisome proliferator-activated receptors. Proc. Natl. Acad. Sci. U. S. A., 98, 13919–13924 (2001).
Zhang, H., Zhang, A., Kohan, D. E., Nelson, R. D., Gonzalez, F. J., and Yang, T., Collecting duct-specific deletion of peroxisome proliferator-activated receptor gamma blocks thiazolidinedione-induced fluid retention. Proc. Natl. Acad. Sci. U. S. A., 102, 9406–9411 (2005).
Zhulun, Y. L., Wang, Z., Motani, A., Mcgee, L. R., Learned, M., Timmermans, P. B., and Chen, J. L., T0903131 (T131); A selective modulator of PPARγ. Diabetes, 53(Suppl 2), A157 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, MK., Chae, Y.N., Kim, H.S. et al. PAR-1622 is a selective peroxisome proliferator-activated receptor γ partial activator with preserved antidiabetic efficacy and broader safety profile for fluid retention. Arch. Pharm. Res. 32, 721–727 (2009). https://doi.org/10.1007/s12272-009-1511-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-009-1511-8