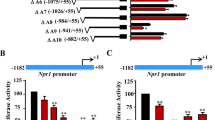
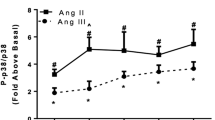
Abstract
The vasopressor octapeptide, angiotensin II (Ang II), exerts homeostatic responses in cardiovascular tissues, including the heart, blood vessel wall, adrenal cortex and liver (a major source of circulating plasma proteins). One of the effects of Ang II is to induce expression of regulatory, structural and cytokine genes that play important roles in long-term control of blood pressure, vascular remodeling, cardiac hypertrophy and inflammation. The identification of nuclear signaling pathways and target transcription factors has provide important insight into cellular responses and the spectrum of genes controlled by Ang II. Here we will review how Ang II activates the transcription factors, Activator Protein 1 (AP-1), Signal Transducer and Activator of Transcription (STATs ), and Nuclear Factor-κB (NF-κB). NF-κB is of particular interest because it is an important mediator of resynthesis of the Ang II precursor, angiotensinogen AGT. Through this positive feedback loop, long-term changes in the activity of the renin angiotensin system occur. Although NF-κB is ubiquitously expressed, surprisingly the mechanism for Ang II-inducible NF-κB regulation differs between aortic smooth muscle cells (VSMCs) and hepatocytes. In VSMC, Ang II induces nuclear translocation of cytoplasmic transactivatory NF-κB proteins through proteolysis of its inhibitor, IkB. By contrast, in hepatocytes, Ang II induces large nuclear isoforms of NF-κB1 to bind DNA through a mechanism independent of changes in IkB turnover. NF-κB activation depends upon the activity of DAG-sensitive PKC isoforms and ROS signaling pathway. These observations indicate that significant differences exist in Ang II signaling depending upon cell-type involved and suggest the possibility that tissue-selective modulation of Ang II effects can be modulated in cardiac tissues.
Similar content being viewed by others
References
Reid I A, Morris BJ, Ganong WG: The renin-angiotensin system. Annu Rev Physiol 40: 377–410, 1978
Corvol P, Jeunemaitre X: Molecular genetics of human hypertension: Role of angiotensinogen. Endo Rev 18: 662–677, 1997
Peach MJ: Renin-angiotensin system: Biochemistry and mechanisms of action. Physiol Rev 57: 313–370, 1977
Walker WG, Whelton PK, Saito H, Russel RP, Hermann J: Relation between blood pressure and renin, renin substrate, AII, aldosterone and urinary sodium and potassium in 574 ambulatory subjects. Hypertension 1: 287–291, 1979
Krakoff LR: Measurement of plasma-renin substrate by radioimmunoassay of angiotensin. I. Concentration in syndromes associated with steroid excess. J Clin Endocrinol Metab 37: 110–117, 1973
Kim HS, Krege JH, Kluckman KD, Hagaman JR, Hodgin JB, Best CF, Jennette JC, Coffman TM, Maeda N, Smithies O: Genetic control of blood pressure and the angiotensinogen locus. Proc Natl Acad Sci USA 92: 2735–2739, 1995
Fukamizu A, Sugimura K, Takimoto E, Sugiyama F, Seo MS, Takahashi S, Hatae T, Kajiwara N, Yagami K, Murakami K: Chimeric renin-angiotensin system demonstrates sustain increase in blood pressure of transgenic mice carrying both human renin and human angiotensinogen genes. J Biol Chem 268: 11617–11621, 1993
Jeunemaitre X, Soubrier F, Kotelevtsev YV, Lifton RP, Williams CS, Charru A, Hunt SC, Hopkins PN, Williams RR, Lalouel JM: Molecular basis of human hypertension: Role of angiotensinogen. Cell 71: 169–180, 1992
Caulfield M, Lavender P, Farrall M, Munroe P, Lawson M, Turner P, Clark AJ: Linkage of the angiotensinogen gene to essential hypertension. New Engl J Med 330: 1629–1633, 1994
Morgan T, Craven C, Nelson L, Lalouel JM, Ward K: Angiotensinogen T235 expression is elevated in decidual spiral arteries. J Clin Invest 100: 1406–1415, 1997
Ward K, Hata A, Jeunemaitre X, Helin L, Nelson C, Namikawa C, Farrington PF, Ogasawara M, Suzumori K, Tomoda S: A molecular variant of angiotensinogen associated with preeclampsia. Nature Genet 4: 59–61, 1998
Hopkins PN, Lifton RP, Hollenberg SM, Jeunemaitre X, Hallouin MC, Skuppin J, Williams CS, Dluhy RG, Lalouel JM, Williams RR, Williams GH: Blunted renal vascular response to angiotensin II is associated with a common variant of the angiotensinogen gene and obesity. J Hypertens 14: 199–207, 1996
Brasier AR, Li J: Mechanisms for inducible control of angiotensinogen gene transcription. J Hypertens 27: 465–475, 1996
Campbell DJ: Circulating and tissue angiotensin systems. J Clin Invest 79: 1–6, 1987
McGehee RE Jr, Ron D, Brasier AR, Habener JF: Differentiation-specific element: A cis-acting developmental switch required for the sustained transcriptional expression of the angiotensinogen gene during hormonal-induced differentiation of 3T3-L1 fibroblasts to adipocytes. Mol Endocrinol 7: 551–560, 1993
Cassis LA, Saye J, Peach MJ: Localization and regulation of rat angiotensinogen messenger RNA. Hypertension 11: 591–596, 1988
Campbell DJ, Habener JF: Angiotensinogen gene is expressed and differentially regulated in multiple tissues of the rat. J Clin Invest 78: 31–39, 1986
Ganten D, Hermann K, Bayer C, Unger T, Lang RE: Angiotensin synthesis in the brain and increased turnover in hypertensive rats. Science 221: 869–871, 1983
Sernia C: Location and secretion of brain angiotensinogen. Reg Pep 57: 1–18, 1995
Celio MR, Inagami T: Angiotensin II immunoreactivity coexists with renin in the juxtaglomerular granular cells of the kidney. Proc Natl Acad Sci USA 78: 3897–3900, 1981
Ingelfinger JR, Zuo W, Fon EA, Ellison KE, Dzau V: In situ hybridization evidence for angiotensinogen messenger RNA in the rat proximal tubule. An hypothesis for the intrarenal renin angiotensin system. J Clin Invest 85: 417–423, 1990
Aguilera G, Schirar A, Baukal A, Catt KJ: Circulating angiotensin II and adrenal receptors after nephrectomy. Nature (London) 289: 507–509, 1981
Racz K, Pinet F, Gasc J-M, Guyene T-T, Corvol P: Coexpression of renin, angiotensiongen and their messenger ribonuclei acids in adrenal tissues. J Clin Endocrinol Metab 75: 730–737, 1992
Swales JD, Abramovici D, Beck F, Bing RF, Loudon M, Thurston H: Arterial wall renin. J Hypertens 1(suppl 1): 17–22, 1983
Naftilan A, Zuo W, Ingelfinger JR, Ryan TJ, Pratt RE, Dzau V: Localization and differential regulation of angiotensinogen mRNA expression in the vascular wall. J Clin Invest 87: 1300–1311, 1991
Sadoshima J, Xu Y, Slayter HS, Izumo S: Autocrine release of angiotensin II mediates stretch-induced hypertrophy of cardiac myocytes in vitro. Cell 75: 977–984, 1993
Sadoshima J, Izumo S: Molecular characterization of angiotensin IIinduced hypertrophy of cardiac myocytes and hyperplasia of cardiac fibroblasts. Critical role of the AT1 receptor subtype. Circ Res 73: 413–423, 1993
Ellison KE, Ingelfinger JR, Pivor M, Dzau V: Androgen regulation of rat renal angiotensinogen messenger RNA expression. J Clin Invest 83: 1941–1945, 1989
Kalinyak JE, Perlman AJ: Tissue-specific regulation of angiotensinogen mRNA accumulation by dexamethasone. J Biol Chem 262: 460–464, 1987
Gunther S, Alexander RW, Atkinson WJ, Gimbrone M Jr: Functional angiotensin II receptors in cultured vascular smooth muscle. J Cell Biol 92: 289–298, 1982
Murphy TJ, Alexander RW, Griendling KK, Runge MS, Bernstein KE: Isolation of a cDNA encoding the vascular type-1 angiotensin II receptor. Nature (Lond) 351: 233–236, 1991
Bouscarel B, Blackmore PF, Exton JH: Characterization of the angiotensin II receptor in primary cultures of rat hepatocytes. J Biol Chem 263: 14913–14919, 1988
Tsuchida S, Matsusaka T, Chen X, Okubo S, Niimura F, Nishimura H, Fogo A, Utsunomiya H, Inagami T, Ichikawa I: Murine double nullizygotes of the angiotensin type 1a and 1b receptor genes duplicate severe abnormal phenotypes of angiotensinogen nullizygotes. J Clin Invest 101: 755–760, 1998
Diaz-Laviada I, Larrodera P, Nieto JL, Cornet ME, Diaz-Meco MT, Sanchez MJ, Guddal PH, Johansen T, Haro A, Moscat J: Mechanism of inhibition of adenylate cyclase by phospholipase c-catalyzed hydrolysis of phosphatidylcholine. J Biol Chem 266: 1170–1176, 1991
Maloney JA, Tsygankova O, Szot A, Yang L, Li Q, Williamson JR: Differential translocation of protein kinase C isozymes by phorbol esters, EGF, and ANG II in rat liver WB cells. Am J Physiol 274: C974–C982, 1998
Griendling KK, Ushio-Fukai M, Lassegue B, Alexander RW: Angiotensin II signaling in vascular smooth muscle. Hypertension 29: 366–373, 1997
Griendling KK, Minieri CA, Ollerenshaw JD, Alexander RW: Angiotensin II stimulates NADH and NADPH oxidase activity in cultured vascular smooth muscle cells. Circ Res 74: 1141–1148, 1994
Suh Y-A, Arnold RS: Cell transformation by the superoxide-generating oxidase Mox1. Nature (London) 401: 79–82, 1999
McWhinney CD, Hunt RA, Conrad KM, Dostal DE, Baker KM: The type 1 angiotensin II receptor couples to STAT1 and STAT3 activation through Jak2 kinase in neonatal rat cardiac myocytes. J Mol Cell Cardiol 29: 2513–2524, 1997
Venema RC, Benema VJ, Eaton DC, Marrero MB: Angiotensin II-induced tyrosin phosphorylation of signal transducers and activators of transcription 1 is regulated by Janus-activated kinase 2 and Fynk kinases and mitogen activated protein kinase phosphatase 1. J Biol Chem 273: 30795–30800, 1998
Marrero MB, Schieffer B, Paxton WG, Heerdt L, Berk BC, Delafontaine P, Bernstein KE: Direct stimulation of Jak/STAT pathway by the angiotensin II AT1 receptor. Nature (London) 375: 247–250, 1995
Liao D-F, Monia B, Dean N, Berk BC: Protein kinase C-ζ mediates angiotensin II activation of ERK1/2 in vascular smooth muscle cells. J Biol Chem 272: 6146–6150, 1997
Kojima I, Kojima K, Kreutter D, Rasmussen H: The temporal integration of the aldosterone secretory response to angiotensin occurs via two intracellular pathways. J Biol Chem 259: 14448–14457, 1984
Clark AJ, Balla T, Jones MR, Catt KJ: Stimulation of early gene expression by angiotensin II in bovine adrenal glomerulosa cells: Roles of calcium and protein kinase C. Mol Endocrinol 6: 1889–1898, 1992
Kingston RE, Bunker CA, Imbalzano AN: Repression and activation by multiprotein complexes that alter chromatin structure. Genes Dev 10: 905–920, 1996
Beato M: Chromatin structure and the regulation of gene expression: Remodeling at the MMTV promoter. J Mol Med 74: 711–724, 1996
Brasier AR, Jamaluddin M, Casola A, Duan W, Shen Q, Garofalo R: A promoter recruitment mechanism for TNFα-induced IL-8 transcription in type II pulmonary epithelial cells: Dependence on nuclear abundance of Rel A, NF-κB1 and c-Rel transcription factors. J Biol Chem 273: 3551–3561, 1998
Angel P, Karin M: The role of Jun, Fos and the AP-1 complex in cell proliferation and transformation. Biochim Biophys Acta 1072: 129–157, 1991
Neyses L, Nouskas J, Luyken J, Fronhoffs S, Oberdorf S, Pfeifer U, Williams RS, Sukhatme VP, Vetter H: Induction of immediate-early genes by angiotensin II and endothelin-1 in adult rat cardiomyocytes. J Hypertens 11: 927–934, 1993
Taubman MB, Berk BC, Izumo S, Tsuda T, Alexander RW, Nadal Ginard B: Angiotensin II induces c-fos mRNA in aortic smooth muscle. Role of Ca2+ mobilization and protein kinase C activation. J Biol Chem 264: 526–530, 1989
Naftilan A, Pratt RE, Eldridge CS, Lin HL, Dzau V: Angiotensin II induces c-fos expression in smooth muscle via transcriptional control. Hypertension 13: 706–711, 1989
Gonzalez-Espinosa C, Garcia-Sainz JA: Angiotensin II and active phorbol esters induce proto-oncogene expression in isolated rat hepatocytes. Biochim Biophys Acta 1136: 309–314, 1992
Wolf G, Neilson EG: Angiotensin II induces cellular hypertrophy in cutlured murine proximal tubular cells. Am J Physiol 269: F768–F777, 1999
Ruiz-Ortega M, Egido J: Angiotensin II modulates cell growth-related events and synthesis of matrix proteins in rela interstitial fibroblasts. Kidney Int 52: 1497–1510, 1997
Blume A, Herdegen T, Unger T: Angiotensin peptides and inducible transcription factors. J Mol Med 77: 339–357, 1999
Angel P, Hattori K, Smeal T, Karin M: The jun proto-oncogen is positively autoregulated by its product. Cell 55: 875–885, 1988
Zohn IE, Yu H, Cox AD, Earp HS: Angiotensin II stimulates calciumdependent activation of c-Jun N-terminal kinase. Mol Cell Biol 15: 6160–6168, 1995
Karin M: The regulation of AP-1 activity by mitogen-activated protein kinase. J Biol Chem 270: 16483–16486, 1995
Kudoh S, Komuro I, Mizuno T, Yamazaki T, Zou Y, Shiojima I, Takekoshi N, Yazaki Y: Angiotensin II stimulates c-Jun NH2-terminal kinase in cultured cardiac myocytes of neonatal rats. Circ Res 80: 139–146, 1997
Darnell JE Jr, Kerr, Startk GR: Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science 264: 1415–1421, 1994
Darnell JE Jr: STATS and gene regulation. Science 277: 1630–1635, 1997
Bhat GJ, Thekkumkara TJ, Thomas WG, Conrad KM, Baker KM: Angiotensin II stimulates sis-inducing factor-like DNA binding activity: Evidence that the AT1a receptor activates transcription factor Stat 91 and/or a related protein. J Biol Chem 269: 31443–31449, 1994
Bhat GJ, Baker KM: Angiotensin II stimulates rapid serine phosphorylation of transcription factor STAT3. Mol Cell Biochem 170: 171–176, 1997
Wen Z, Zhong A, Darnell JE Jr: Maximal activation of transcription by Stat1 and Stat3 requires both tyrosine and serine phosphorylation. Cell 82: 241–250, 1995
Pan J, Fukuda K, Kodama H, Makino S, Takashi T, Sano M, Hori S, Ogawa S: Role of angiotensin II in activation of the JAK/STAT pathway induced by acute pressure overload in the rat heart. Circ Res 81: 611–617, 1997
Mascareno E, Dhar M, Siddiqui MAQ: Signal transduction and activator of transcription (STAT) protein-dependent activation of angiotensinogen promoter: A cellular signal for hypertrophy in cardiac muscle. Proc Natl Acad Sci USA 95: 5590–5594, 1998
Patterson C, Ruef J, Madamanchi NR, Barry-Lane P, Hu Z, Horaist C, Ballinger CA, Brasier AR, Bode C, Runge MS: Stimulation of a vascular smooth muscle cell NAD(P)H oxidase by thrombin. Evidence that p47(phox) may participate in forming this oxidase in vitro and in vivo. J Biol Chem 274: 19814–19822, 1999
Brasier AR, Han Y, Sherman CT: Transcriptional regulation of angiotensinogen gene expression. Vit Horm 57: 217–247, 1999
Siebenlist U, Franzoso G, Brown K: Structure, regulation and function of NF-κB. Annu Rev Cell Biol 10: 405–455, 1994
Han Y, Brasier AR: Mechanism for biphasic Rel A: NF-κB1 nuclear translocation in tumor necrosis factor α-stimulated hepatocytes. J Biol Chem 272: 9823–9830, 1997
Maniatis T: Catalysis by a multiprotein IκB kinase complex. Science 278: 818–819, 1997
Chen JZ, Parent L, Maniatis T: Site-specific phosphorylation of IκBa by a novel ubiquitination-dependent protein kinase activity. Cell 84: 853–862, 1996
Palombella VJ, Rando OJ, Goldberg AL, Maniatis T: The ubiquitinproteasome pathway is required for processing the NF-κB1 precursor protein and the activation of NF-κB. Cell 78: 773–785, 1994
Han Y, Weinman SA, Boldogh S, Brasier AR: TNFa-inducible IκBa proteolysis and NF-κB activation mediated by cytosolic m-Calpain. Biol Chem 274: 787–794, 1999
Han Y, Brasier AR: Angiotensin II induces IL-6 transcription in vascular smooth muscle cells through pleiotropic activation of NF-κB transcription factors. Circ Res 84: 695–703, 1999
Bourcier T, Sukhova G, Libby P: The nuclear factor-κB signaling pathway participates in dysregulation of vascular smooth muscle cells in vitro and in human atherosclerosis. J Biol Chem 272: 15817–15824, 1997
Brand K, Page S, Rogler G, Bartsch A, Brandl R, Kneuchel R, Page M, Kaltschmidt C, Baeuerle PA, Neumeier D: Activated transcription factor nuclear factor-κB is present in the atherosclerotic lesion. J Clin Invest 97: 1715–1722, 1996
Brasier AR, Li J, Wimbish KA: Tumor necrosis factor activates angiotensinogen gene expression by the Rel A transactivator. J Hypertens 27: 1009–1017, 1996
Li J, Brasier AR: Angiotensinogen gene activation by AII is mediated by the Rel A (NF-κB p65) transcription factor: One mechanism for the renin angiotensin system (RAS) positive feedback loop in hepatocytes. Mol Endocrinol 10: 252–264, 1996
Akira S, Kishimoto T: IL-6 and NF-IL6 in acute-phase response and viral infection. Immunol Rev 127: 25–50, 1992
Loppnow H, Libby P: Proliferating or interleukin-1 activated human vascular smooth muscle cells secrete copious IL-6. J Clin Invest 85: 731–738, 1990
Gaumond F, Fortin D, Stankova J, Rola-Pleszczynski M: Differential signaling pathways in platelet-activating factor-inducted proliferation and interleukin-6 production by rat vascular smooth muscle cells. J Cardiovasc Pharmacol 30: 169–175, 1997
Beasley D: Phorbol ester and interleukin-1 induce interleukin-6 gene expression in vascular smooth muscle cells via independent pathways. J Cardiovasc Pharmacol 29: 323–330, 1997
Ikeda U, Ikeda M, Oohara T: Interleukin-6 stimulates c-myc expression and proliferation of cultured vascular smooth muscle cells. Am J Physiol 260: H1713–H1717, 1991
Yasukawa K, Hirano T, Watanabe Y: Structure and expression of human B-cell stimulatory factor 2 (BSF-2) gene. EMBO J 6: 2939, 1987
Loppnow H, Bil R, Hirt S, Schonbeck W, Herzberg M, Werdan K, Rietschel ET, Brandt E, Flad HD: Platelet derived IL-1 induces cytokine production, but not proliferation of human vascular smooth muscle cells. Blood 91: 134–141, 1998
Vlahopoulos S, Boldogh I, Brasier AR: NF-κB dependent induction of interleukin-8 gene expression by tumor necrosis factor a: Evidence for an antioxidant sensitive activating pathway distinct from nuclear translocation. Blood 94: 1878–1889, 1999
Kranzhofer R, Schmidt J, Pfeiffer CAH, Hagl S, Libby P, Kubler W: Angiotensin induces inflammatory activation of human vascular smooth muscle cells. Arteriosc Thromb Vasc Biol 19: 1623–1629, 1999
Hilgenfeldt U, Schwind S: Angiotensin II is the mediator of the increase in hepatic angiotensinogen synthesis after bilateral nephrectomy. Am J Physiol 265: E414–E418, 1993
Nakamura A, Iwao H, Fukui K, Kimura S, Tamaki T, Nakanishi S, Abe Y: Regulation of liver angiotensinogen and kidney renin mRNA levels by angiotensin II. Am J Physiol 258: E1–E6, 1990
Kohara K, Brosnihan KB, Ferrario CM, Milsted A: Peripheral and central angiotensin II regulates expression of genes of the renin-angiotensin system. Am J Physiol 262: E651–E657, 1992
Eggena P, Zhu JH, Clegg K, Barrett JD: Nuclear angiotensin receptors induce transcription of renin and angiotensinogen mRNA. Hypertension 22: 496–501, 1993
Jamaluddin M, Meng T, Sun J, Boldogh S, Han Y, Brasier AR: Angiotensin II induces constitutive Nuclear factor-κB1 (NF-κB1) isoforms to bind the angiotensinogen gene acute phase response element: A stimulus-specific pathway for NF-κB activation in hepatocytes. Mol Endocrinol 14: 99–113, 2000
Grumont RJ, Fecondo J, Gerondakis S: Alternate RNA splicing of murine nfkb1 generates a nuclear isoform of the p50 precursor NF-κB1 that can function as a transactivator of NF-κB-regulated transcription. Mol Cell Biol 14: 8460–8470, 1994
Wadgaonkar R, Phelps KM, Haque Z, Williams AJ, Silverman ES, Collins T: CREB binding protein is a nuclear integrator of nuclear factor-κB and p53 signaling. J Biol Chem 274: 1879–1882, 1999
Perkins ND, Felzien LZ, Betts JC, Leung K, Beach DH, Nabel GJ: Regulation of NF-κB by cyclin-dependent kinases associated with the p300 coactivator. Science 275: 523–527, 1997
Khan AU, Wilson T: Reactive oxygen species as cellular messengers. Chem Biol 2: 437, 1995
Sundaresan M, Yu Z-X, Ferrans V, Irani K, Finkel T: Requirement for generation of H202 for platelet-derived growth factor signal transduction. Science 270: 296–299, 1995
Lo YY, Cruz TF: Involvement of reactive oxygen species in cytokine and growth factor induction of c-fos expression in chrondrocytes. J Biol Chem 270: 11727–11730, 1995
Rao GN, Berk BC: Circ Res 70: 593–599, 1992
Ginn-Pease ME, Whisler RL: Redox signals and NF-κB activation in T cells. Free Rad Biol Med 25: 346–1998
Alexander RW: Hypertension and the pathogenesis of atherosclerosis. Oxidative stress and the mediation of arterial inflammatory response: A new perspective. J Hypertens 25: 155–161, 1995
Ross R: The pathogenesis of atherosclerosis: A perspective for the 1990s. Nature (London) 362: 801–809, 1993
Kume N, Cybulsky MI, Gimbrone MA Jr: Lysophosphatidylcholine, a component of atherogenic lipoproteins, induces mononuclear leukocyte adhesion molecules in cultured human and rabbit arterial endothelial cells. J Clin Invest 90: 1138–1144, 1992
Han Y, Meng T, Murray NR, Fields AP, Brasier AR: IL-1 Induced NFκB-IκBα autoregulatory feedback loop in hepatocytes: A role for PKCα in post-transcriptional regulation of IκBα resynthesis. J Biol Chem 274: 939–947, 1999
Naftilan AJ, Pratt RE, Dzau VJ: Induction of platelet-derived growth factor A-chain and c-myc gene expressions by angiotensin II in cultured rat vascular smooth muscle cells. J Clin Invest 83: 1419–1424, 1989
Pastore L, Tessitore A, Martinotti S, Toniato E, Alesse E, Bravi MC, Ferri C, Desideri G, Gulino A, Santucci A: Angiotensin II stimulates intercellular adhesion molecule-1 (ICAM-1) expression by human vascular endothelial cells and increases soluble ICAM-1 release in vivo. Circulation 100: 1646–1652, 1999
Tummala PE, Chen XL, Sundell CL, Laursen JB, Hammes CP, Alexander RW, Harrison DG, Medford RM: Angiotensin II induces vascular cell adhesion molecule-1 expression in rat vasculature: A potential link between the renin-angiotensin system and atherosclerosis. Circulation 100: 1223–1229, 1999
Wegenka UM, Buschmann J, Lutticken C, Heinrich PC, Horn F: Acutephase response factor, a nuclear factor binding to acute-phase response elements, is rapidly activated by interleukin-6 at the postranslational level. Mol Cell Biol 13: 276–288, 1993
Hocke GM, Barry D, Fey GH: Synergistic action of interleukin-6 and glucocorticoids is mediated by the interleukin-6 response element of the rat a2-macroglobulin gene. Mol Cell Biol 12: 2282–2294, 1992
Akira S, Nishio Y, Inoue M, Wang X-J, Wei S, Matsusaka T, Yoshida K, Sudo T, Naruto M, Kishimoto T: Molecular cloning of APRF, a novel IFN-stimulated gene factor 3 p91-related transcription factor involved in the gp130-mediated signaling pathway. Cell 63-71, 1994
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Brasier, A.R., Jamaluddin, M., Han, Y. et al. Angiotensin II induces gene transcription through cell-type-dependent effects on the nuclear factor-κB (NF-κB) transcription factor. Mol Cell Biochem 212, 155–169 (2000). https://doi.org/10.1023/A:1007133710837
Issue Date:
DOI: https://doi.org/10.1023/A:1007133710837