Abstract
The retinoblastoma tumour suppressor (Rb) pathway is believed to have a critical role in the control of cellular proliferation by regulating E2F activities1,2. E2F1, E2F2 and E2F3 belong to a subclass of E2F factors thought to act as transcriptional activators important for progression through the G1/S transition3. Here we show, by taking a conditional gene targeting approach, that the combined loss of these three E2F factors severely affects E2F target expression and completely abolishes the ability of mouse embryonic fibroblasts to enter S phase, progress through mitosis and proliferate. Loss of E2F function results in an elevation of p21Cip1 protein, leading to a decrease in cyclin-dependent kinase activity and Rb phosphorylation. These findings suggest a function for this subclass of E2F transcriptional activators in a positive feedback loop, through down-modulation of p21Cip1, that leads to the inactivation of Rb-dependent repression and S phase entry. By targeting the entire subclass of E2F transcriptional activators we provide direct genetic evidence for their essential role in cell cycle progression, proliferation and development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Dyson, N. The regulation of E2F by pRB-family proteins. Genes Dev. 12, 2245–2262 (1998).
Nevins, J. R. Toward an understanding of the functional complexity of the E2F and retinoblastoma families. Cell Growth Differ. 9, 585–593 (1998).
DeGregori, J., Leone, G., Miron, A., Jakoi, L. & Nevins, J. R. Distinct roles for E2F proteins in cell growth control and apoptosis. Proc. Natl Acad. Sci. USA 94, 7245–7250 (1997).
Duronio, R. J., O'Farrell, P. H., Xie, J E., Brook, A. & Dyson, N. The transcription factor E2F is required for S phase during Drosophila embryogenesis. Genes Dev. 9, 1445–1455 (1995).
Royzman, I., Whittaker, A. J. & Orr-Weaver, T. L. Mutations in Drosophila DP and E2F distinguish G1–S progression from an associated transcriptional program. Genes Dev. 11, 1999–2011 (1997).
Field, S. J. et al. E2F-1 functions in mice to promote apoptosis and suppress proliferation. Cell 85, 549–561 (1996).
Humbert, P. O. et al. E2F4 is essential for normal erythrocyte maturation and neonatal viability. Mol. Cell 6, 281–291 (2000).
Humbert, P. O. et al. E2f3 is critical for normal cellular proliferation. Genes Dev. 14, 690–703 (2000).
Leone, G. et al. Myc requires distinct E2F activities to induce S phase and apoptosis. Mol. Cell 8, 105–113 (2001).
Lindeman, G. J. et al. A specific, nonproliferative role for E2F-5 in choroid plexus function revealed by gene targeting. Genes Dev. 12, 1092–1098 (1998).
Rempel, R. E. et al. Loss of E2F4 activity leads to abnormal development of multiple cellular lineages. Mol. Cell. 6, 293–306 (2000).
Yamasaki, L. et al. Tumor induction and tissue atrophy in mice lacking E2F-1. Cell 85, 537–548 (1996).
Lees, J. A. et al. The retinoblastoma protein binds to a family of E2F transcription factors. Mol. Cell. Biol. 13, 7813–7825 (1993).
Leone, G. et al. E2F3 activity is regulated during the cell cycle and is required for the induction of S phase. Genes Dev. 12, 2120–2130 (1998).
Leone, G. et al. Identification of a novel E2F3 product suggests a mechanism for determining specificity of repression by Rb proteins. Mol. Cell. Biol. 20, 3626–3632 (2000).
Lukas, C. et al. Accumulation of cyclin B1 requires E2F and cyclin-A-dependent rearrangement of the anaphase-promoting complex. Nature 401, 815–881 (1999).
Ishida, S. et al. Role for E2F in control of both DNA replication and mitotic functions as revealed from DNA microarray analysis. Mol. Cell. Biol. 21, 4684–4699 (2001).
Muller, H. et al. E2Fs regulate the expression of genes involved in differentiation, development, proliferation, and apoptosis. Genes Dev. 15, 267–285 (2001).
Brehm, A. et al. Retinoblastoma protein recruits histone deacetylase to repress transcription. Nature 391, 597–601 (1998).
Harbour, J. W. & Dean, D. C. Chromatin remodeling and Rb activity. Curr. Opin. Cell Biol. 12, 685–689 (2000).
Luo, R. X., Postigo, A. A. & Dean, D. C. Rb interacts with histone deacetylase to repress transcription. Cell 92, 463–473 (1998).
Magnaghi-Jaulin, L. et al. Retinoblastoma protein represses transcription by recruiting a histone deacetylase. Nature 391, 601–605 (1998).
Weintraub, S. J., Prater, C. A. & Dean, D. C. Retinoblastoma protein switches the E2F site from positive to negative element. Nature 358, 259–261 (1992).
Weintraub, S. J. et al. Mechanism of active transcriptional repression by the retinoblastoma protein. Nature 375, 812–815 (1995).
Zhang, H. S., Postigo, A. A. & Dean, D. C. Active transcriptional repression by the Rb–E2F complex mediates G1 arrest triggered by p16INK4a, TGFβ, and contact inhibition. Cell 97, 53–61 (1999).
Tsai, K. Y. et al. Mutation of E2f-1 suppresses apoptosis and inappropriate S phase entry and extends survival of Rb-deficient mouse embryos. Mol. Cell 2, 293–304 (1998).
Yamasaki, L. et al. Loss of E2F-1 reduces tumorigenesis and extends the lifespan of Rb1(+/-) mice. Nature Genet. 18, 360–364 (1998).
Ziebold, U., Reza, T., Caron, A. & Lees, J. A. E2F3 contributes both the inappropriate proliferation and to the apoptosis arising in Rb mutant embryos. Genes Dev. 15, 386–391 (2001).
Pear, W. S., Nolan, G. P., Scott, M. L. & Baltimore, D. Production of high-titer helper-free retroviruses by transient transfection. Proc. Natl Acad. Sci. USA 90, 8392–8396 (1993).
Nevins, J. R., DeGregori, J., Jakoi, L. & Leone, G. in Methods in Enzymology (ed. Dunphy, W. G.) 678 (Academic, San Diego, 1997).
Acknowledgements
We are grateful to R. Premont for providing the loxP vectors. We thank C. Bock for assistance in generating the E2F3 chimaeric mice; C. Brown and L. Jakoi for various technical assistance; and M. Weinstein for critical comments on the manuscript. This work was supported by grants from the National Institutes of Health (NIH) (G.L.). L.W. was supported by an NIH award, C.T. was supported by the Up on the Roof Human Cancer Genetics Posdoctoral Fellowship, J.R.N. is an investigator of the Howard Hughes Medical Institute, and G.L. is a V-Foundation and Pew Charitable Trust Scholar.
Author information
Authors and Affiliations
Corresponding author
Supplementary information

Supplementary Table 1
(GIF 3.18 KB)
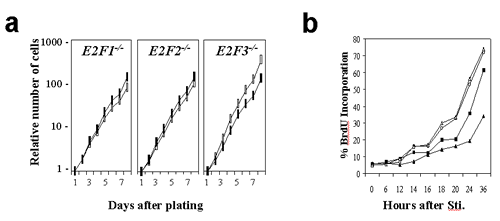
Supplementary Figure 1
(GIF 11.6 KB)
Loss of E2F3 leads to defects in cellular proliferation and S-phase entry. Growth curves (a) and BrdU incorporation assays (b) of E2F-deficient primary MEFs (closed symbols) in comparison with their wild-type counterparts (open symbols).
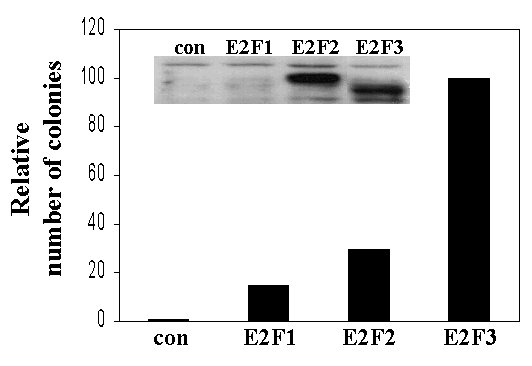
Supplementary Figure 2
(GIF 8.03 KB)
Ectopic expression of E2F1, E2F2, or E2F3 can rescue the proliferation defects of TKO cells. Colony formation assays of E2F1-/-E2F2-/-E2F3f/f MEFs infected with the indicated retrovirus as well as pBpuro-Cre. (con): pBhygro; (E2F1, E2F2, E2F3): pBhygro-HA-tagged E2F1, E2F2, and E2F3a, respectively. Similar results were obtained in four independent experiments; a representative experiment is shown. (inset) Western blot using an antibody against HA-epitope.

Supplementary Figure 3
(JPG 23 KB)
Northern blot analysis of various E2F-responsive genes in E2F3f/f (a) or E2F2-/-E2F3f/ (b) MEFs that were infected with a control or a Cre-retrovirus. The experiment was performed as described in Figure 3a of the main text. (con): pBhygro; (E2F3a): pBhygro-myc-tagged E2F3a. c, Deletion of E2F1, E2F2, and E2F3 does not affect early mitogenic events. Western blot of retrovirus-infected E2F1-/-E2F2-/-E2F3f/f MEFs using antibodies against phosphorylated forms of Erk1/2 or Akt; the same blots were re-probed with antibodies against total Erk1/2 and Akt to verify equal loading (data not shown).
Rights and permissions
About this article
Cite this article
Wu, L., Timmers, C., Maiti, B. et al. The E2F1–3 transcription factors are essential for cellular proliferation. Nature 414, 457–462 (2001). https://doi.org/10.1038/35106593
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35106593
This article is cited by
-
Synthetic lethality in large-scale integrated metabolic and regulatory network models of human cells
npj Systems Biology and Applications (2023)
-
Prediction of human protein interactome of dengue virus non-structural protein 5 (NS5) and its downstream immunological implications
3 Biotech (2023)
-
New roles for AP-1/JUNB in cell cycle control and tumorigenic cell invasion via regulation of cyclin E1 and TGF-β2
Genome Biology (2022)
-
The miR-532-E2F1 feedback loop contributes to gastric cancer progression
Cell Death & Disease (2022)
-
Gene regulatory network analysis defines transcriptome landscape with alternative splicing of human umbilical vein endothelial cells during replicative senescence
BMC Genomics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.



