Abstract
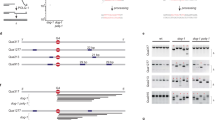
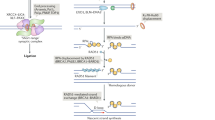
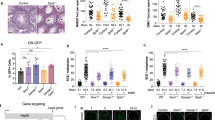
Double-strand breaks occur during DNA replication and are also induced by ionizing radiation. There are at least two pathways which can repair such breaks: non-homologous end joining and homologous recombination (HR). Although these pathways are essentially independent of one another, it is possible that the proteins Mre11, Rad50 and Xrs2 are involved in both pathways in Saccharomyces cerevisiae1. In vertebrate cells, little is known about the exact function of the Mre11–Rad50–Nbs1 complex in the repair of double-strand breaks because Mre11- and Rad50-null mutations are lethal2. Here we show that Nbs1 is essential for HR-mediated repair in higher vertebrate cells. The disruption of Nbs1 reduces gene conversion and sister chromatid exchanges, similar to other HR-deficient mutants3. In fact, a site-specific double-strand break repair assay showed a notable reduction of HR events following generation of such breaks in Nbs1-disrupted cells. The rare recombinants observed in the Nbs1-disrupted cells were frequently found to have aberrant structures, which possibly arise from unusual crossover events, suggesting that the Nbs1 complex might be required to process recombination intermediates.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Haber, J. E. The many interfaces of Mre11. Cell 95, 583–586 (1998)
Yamaguchi-Iwai, Y. et al. Mre11 is essential for the maintenance of chromosomal DNA in vertebrate cells. EMBO J. 18, 6619–6629 (1999)
Sonoda, E. et al. Sister chromatid exchanges are mediated by homologous recombination in vertebrate cells. Mol. Cell. Biol. 19, 5166–5169 (1999)
White, C. I. & Haber, J. E. Intermediates of recombination during mating type switching in Saccharomyces cerevisiae. EMBO J. 9, 663–673 (1990)
Furuse, M. et al. Distinct roles of two separable in vitro activities of yeast mre11 in mitotic and meiotic recombination. EMBO J. 17, 6412–6425 (1998)
Moreau, S., Ferguson, J. R. & Symington, L. S. The nuclease activity of Mre11 is required for meiosis but not for mating type switching, end joining, or telomere maintenance. Mol. Cell. Biol. 19, 556–566 (1999)
Paull, T. T. & Gellert, M. The 3′ to 5′ exonuclease activity of Mre11 facilitates repair of DNA double-strand breaks. Mol. Cell 1, 969–979 (1998)
Paull, T. T. & Gellert, M. Nbs1 potentiates ATP-driven DNA unwinding and endonuclease cleavage by the Mre11/Rad50 complex. Genes Dev. 13, 1276–1288 (1999)
Trujillo, K. M., Yuan, S. S., Lee, E. Y. & Sung, P. Nuclease activities in a complex of human recombination and DNA repair factors Rad50, Mre11, and p95. J. Biol. Chem. 273, 21447–21450 (1998)
Usui, T. et al. Complex formation and functional versatility of Mre11 of budding yeast in recombination. Cell 95, 705–716 (1998)
Tavassoli, M., Shayeghi, M., Nasim, A. & Watts, F. Z. Cloning and characterization of the Schizosaccharomyces pombe rad32 gene: a gene required for repair of double strand breaks and recombination. Nucleic Acids Res. 23, 383–388 (1995)
Petrini, J. H. et al. Isolation and characterization of the human MRE11 homologue. Genomics 29, 80–86 (1995)
Carney, J. P. et al. The hMre11/hRad50 protein complex and Nijmegen breakage syndrome: linkage of double-strand break repair to the cellular DNA damage response. Cell 93, 477–486 (1998)
Matsuura, S. et al. Positional cloning of the gene for Nijmegen breakage syndrome. Nature Genet. 19, 179–181 (1998)
Varon, R. et al. Nibrin, a novel DNA double-strand break repair protein, is mutated in Nijmegen breakage syndrome. Cell 93, 467–476 (1998)
Zhu, J., Petersen, S., Tessarollo, L. & Nussenzweig, A. Targeted disruption of the Nijmegen breakage syndrome gene NBS1 leads to early embryonic lethality in mice. Curr. Biol. 11, 105–109 (2001)
Buerstedde, J. M. & Takeda, S. Increased ratio of targeted to random integration after transfection of chicken B cell lines. Cell 67, 179–188 (1991)
Maser, R. S., Zinkel, R. & Petrini, J. H. J. An alternative mode of translation permits production of a variant NBS1 protein from the common Nijmegen breakage syndrome allele. Nature Genet. 27, 417–421 (2001)
Grenon, M., Gilbert, C. & Lowndes, N. F. Checkpoint activation in response to double-strand breaks requires the Mre11/Rad50/Xrs2 complex. Nature Cell Biol. 3, 844–847 (2001)
D'Amours, D. & Jackson, S. P. The yeast Xrs2 complex functions in S phase checkpoint regulation. Genes Dev. 15, 2238–2249 (2001)
Bezzubova, O. Y., Silbergleit, A., Yamaguchi-Iwai, Y., Takeda, S. & Buerstedde, J. M. Reduced X-ray resistance and homologous recombination frequencies in a RAD54-/- mutant of the chicken DT40 cell line. Cell 89, 185–193 (1997)
Hasty, P., Rivera-Perez, J., Chang, C. & Bradley, A. Target frequency and integration pattern for insertion and replacement vectors in embryonic stem cells. Mol. Cell. Biol. 11, 4509–4517 (1991)
de Jager, M. et al. DNA-binding and strand-annealing activities of human Mre11: implications for its roles in DNA double-strand break repair pathways. Nucleic Acids Res. 29, 1317–1325 (2001)
Johnson, R. D., Liu, N. & Jasin, M. Mammalian XRCC2 promotes the repair of DNA double-strand breaks by homologous recombination. Nature 401, 397–399 (1999)
Holliday, R. A mechanism for gene conversion in fungi. Genet. Res. 5, 282–304 (1964)
Johnson, R. D. & Jasin, M. Sister chromatid gene conversion is a prominent double-strand break repair pathway in mammalian cells. EMBO J. 19, 3398–3407 (2000)
Tauchi, H. et al. The forkhead-associated domain of NBS1 is essential for nuclear foci formation after irradiation, but not essential for hRAD50/hMRE11/NBS1 complex DNA repair activity. J. Biol. Chem. 276, 12–15 (2001)
Sugawara, H., Kurosaki, M., Takata, M. & Kurosaki, T. Genetic evidence for involvement of type 1, type 2 and type 3 inositol 1,4,5-trisphosphate receptors in signal transduction through the B-cell antigen receptor. EMBO J. 16, 3078–3088 (1997)
van Gent, D. C., Ramsden, D. A. & Gellert, M. The RAG1 and RAG2 proteins establish the 12/23 rule in V(D)J recombination. Cell 85, 107–113 (1996)
Verkaik, N. S. et al. Different types of V(D)J recombination and end-joining defects in DNA double-strand break repair mutant mammalian cells. Eur. J. Immunol. (in the press)
Acknowledgements
We thank L. N. Kapp for helping to prepare the manuscript; M. Jasin for providing the SCneo construct; A. Shinohara for anti-Rad51 antibody. We also thank H. Komata, C. Muranaka, T. Jo, M. Ueda, A. Kodama-Kamesako and A. Okamoto for laboratory assistance. This work was supported in part by the Ministry of Education, Science, Sports and Culture of Japan (H.T. and K.K.), and by the Nuclear Safety Research Association of Japan (H.T.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Tauchi, H., Kobayashi, J., Morishima, Ki. et al. Nbs1 is essential for DNA repair by homologous recombination in higher vertebrate cells. Nature 420, 93–98 (2002). https://doi.org/10.1038/nature01125
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature01125
This article is cited by
-
DNA double-strand break genetic variants in patients with premature ovarian insufficiency
Journal of Ovarian Research (2023)
-
MRE11 promotes oral cancer progression through RUNX2/CXCR4/AKT/FOXA2 signaling in a nuclease-independent manner
Oncogene (2021)
-
DNA repair- and nucleotide metabolism-related genes exhibit differential CHG methylation patterns in natural and synthetic polyploids (Brassica napus L.)
Horticulture Research (2021)
-
NBS1 I171V variant underlies individual differences in chromosomal radiosensitivity within human populations
Scientific Reports (2021)
-
PP4 deficiency leads to DNA replication stress that impairs immunoglobulin class switch efficiency
Cell Death & Differentiation (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.