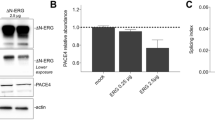
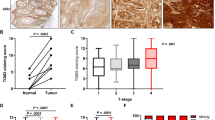
Summary
CPT-11 is a prodrug activated by carboxylesterases to the active metabolite SN-38 which is a potent inhibitor of topoisomerase I. CPT-11 is of clinical interest in the treatment of colorectal cancer. We evaluated the activities of CPT-11 converting carboxylesterase (CPT-CE) and topoisomerase I (topo I) in 53 colorectal tumours, in eight liver metastases and in normal tissue adjacent to the tumours. Both CPT-CE and topo I activities were widely variable in the malignant and the normal tissue of patients with colorectal carcinomas. CPT-CE was only two to threefold lower in primary tumours compared to normal liver, suggesting that a local conversion to SN-38 might occur in tumour cells. CPT-CE was similar in liver and in normal colon tissues. Levels of topo I in tumour ranged from 580 to 84 900 U mg protein–1 and was above 40 000 U mg protein–1 in 11 of 53 patients. Similarly, a very high ratio (> 5) between tumour and normal tissues were observed in 12 of 53 patients. An inverse correlation was observed between the topo I activity and the clinical stage of disease. Clinical studies are in progress in our institution to explore a possible relationship between CPT-CE and topo I activities in tumour cells and the response to CPT-11-based chemotherapy in patients with colorectal cancer.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Benedetti, P., Fiorani, P., Capuani, L. & Wang, J. C. (1993). Camptothecin resistance from a single mutation changing glycine 363 of human DNA topoisomerase I to cysteine. Cancer Res 53: 4343–4348.
Bradford, M. M. (1976). A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248–254.
Bronstein, I. B., Vorobyev, S., Timofeev, A., Jolles, C. J., Alder, S. I. & Holden, J. A. (1996). Elevations of DNA topoisomerase I catalytic activity and immunoprotein in human malignancies. Oncol Res 8: 17–25.
Codegoni, A. M., Castagna, S., Mangioni, C., Scovassi, A. I., Broggini, M. & D’Incalci, M. (1998). DNA-topoisomerase I activity and content in epithelial ovarian cancer. Ann Oncol 9: 313–319.
Cornarotti, M., Capranico, G., Bohm, S., Oriana, S., Spatti, G. B., Mariani, L., Ballabio, G. & Zunino, F. (1996). Gene expression of DNA topoisomerases I, II alpha and II beta and response to cisplatin-based chemotherapy in advanced ovarian carcinoma. Int J Cancer 67: 479–484.
Creemers, G. J., Lund, B. & Verweij, J. (1994). Topoisomerase I inhibitors: topotecan and irenotecan. Cancer Treat Rev 20: 73–96.
Danks, M. K., Morton, C. L., Pawlik, C. A. & Potter, P. M. (1998). Overexpression of a rabbit liver carboxylesterase sensitizes human tumor cells to CPT-11. Cancer Res 58: 20–22.
Deffie, A. M., Batra, J. K. & Goldenberg, G. J. (1989). Direct correlation between DNA topoisomerase II activity and cytotoxicity in adriamycin-sensitive and -resistant P388 leukemia cell lines. Cancer Res 49: 58–62.
Giovanella, B. C., Stehlin, J. S., Wall, M. E., Wani, M. C., Nicholas, A. W., Liu, L. F., Silber, R. & Potmesil, M. (1989). DNA topoisomerase I-targeted chemotherapy of human colon cancer in xenografts. Science 246: 1046–1048.
Gupta, E., Lestingi, T. M., Mick, R., Ramirez, J., Vokes, E. E. & Ratain, M. J. (1994). Metabolic fate of irinotecan in humans: correlation of glucuronidation with diarrhea. Cancer Res 54: 3723–3725.
Haaz, M. C., Rivory, L., Riché, C., Vernillet, L. & Robert, J. (1998). Metabolism of irinotecan (CPT-11) by human hepatic microsomes: participation of cytochrome P-450 3A and drug interactions. Cancer Res 58: 468–472.
Hosokawa, M., Maki, T. & Satoh, T. (1990). Characterization of molecular species of liver microsomal carboxylesterases of several animal species and humans. Arch Biochem Biophys 277: 219–227.
Husain, I., Mohler, J. L., Seigler, H. F. & Besterman, J. M. (1994). Elevation of topoisomerase I messenger RNA, protein, and catalytic activity in human tumors: demonstration of tumor-type specificity and implications for cancer chemotherapy. Cancer Res 54: 539–546.
Jansen, W. J. M., Zwart, B., Hulscher, S. T. M., Giaccone, G., Pinedo, H. M. & Boven, E. (1997). CPT-11 in human colon-cancer cell lines and xenografts – characterization of cellular sensitivity determinants. Int J Cancer 70: 335–340.
Jaxel, C., Kohn, K. W., Wani, M. C., Wall, M. E. & Pommier, Y. (1989). Structure-activity study of the action of camptothecin derivatives on mammalian topoisomerase I: evidence for a specific receptor site and relation to antitumor activity. Cancer Res 49: 1465–1469.
Kawato, Y., Furuta, T., Aonuma, M., Yasuoka, M., Yokokura, T. & Matsumoto, K. (1991). Antitumor activity of a camptothecin derivative, CPT-11, against human tumor xenografts in nude mice. Cancer Chemother Pharmacol 28: 192–198.
Knab, A. M., Fertala, J. & Bjornsti, M. A. (1993). Mechanisms of camptothecin resistance in yeast DNA topoisomerase I mutants. J Biol Chem 268: 22322–22330.
Kojima, A., Hackett, N. R., Ohwada, A. & Crystal, R. G. (1998). In vivo human carboxylesterase cDNA gene transfer to activate the prodrug CPT-11 for local treatment of solid tumors. J Clin Invest 101: 1789–1796.
Kunimoto, T., Nitta, K., Tanaka, T., Uehara, N., Baba, H., Takeuchi, M., Yokokura, T., Sawada, S., Miyasaka, T. & Mutai, M. (1987). Antitumor activity of 7-ethyl-10-[4-(1-piperidino)-1-piperidino]carbonyloxy-camptothecin, a novel water-soluble derivative of camptothecin, against murine tumors. Cancer Res 47: 5944–5947.
Lund-Pero, M., Jeppson, B. & Pero, R. (1994). Reduced non-specific steroidal esterase activity in human malignant tumor tissue from liver, colon and breast when compared to peritumoral and normal tissue levels. Anticancer Res 14: 2747–2754.
McKay, J. A., Murray, G. I., Weaver, R. J., Even, S. W. B., Melvin, W. T. & Burke, M. D. (1993). Xenobiotic metabolising enzyme expression in colonic neoplasia. Gut 34: 1234–1239.
McLeod, H. L., Douglas, F., Oates, M., Symonds, R. P., Prakash, D., van der Zee, A. G., Kaye, S. B., Brown, R. & Keith, W. N. (1994). Topoisomerase I and II activity in human breast, cervix, lung and colon cancer. Int J Cancer 59: 607–611.
Massaad, L., de Waziers, I., Ribrag, V., Janot, F., Beaune, P. H., Morizet, J., Gouyette, A. & Chabot, G. G. (1993). Comparison of mouse and human colon tumors with regard to phase I and phase II drug-metabolizing enzyme systems. Cancer Res 52: 6567–6575.
Miller, S. B., Main, A. R. & Rush, R. S. (1980). Purification and physical properties of oligomeric and monomeric carboxylesterases from rabbit liver. J Biol Chem 255: 7161–7167.
Niimi, S., Nakagawa, K., Sugimoto, Y., Nishio, K., Fujiwara, Y., Yokoyama, S., Terashima, Y. & Saijo, N. (1992). Mechanism of cross-resistance to a camptothecin analogue (CPT-11) in a human ovarian cancer cell line selected by cisplatin. Cancer Res 52: 328–333.
Ogasawara, H., Nishio, K., Kanzawa, F., Lee, Y. S., Funayama, Y., Ohira, T., Kuraishi, Y., Isogai, Y. & Saijo, N. (1995). Intracellular carboxyl esterase activity is a determinant of cellular sensitivity to the antineoplastic agent KW-2189 in cell lines resistant to cisplatin and CPT-11. Jpn J Cancer Res 86: 124–129.
Potmesil, M., Hsiang, Y. H., Liu, L. F., Bank, B., Grossberg, H., Kirschenbaum, S., Forlenza, T. J., Penziner, A., Kanganis, D., Knowles, D., Traganos, F. & Silber, R. (1988). Resistance of human leukemic and normal lymphocytes to drug-induced DNA cleavage and low levels of DNA topoisomerase II. Cancer Res 48: 3537–3543.
Rivory, L. P. & Robert, J. (1994). Reversed-phase high-performance liquid chromatographic method for the simultaneous quantitation of the carboxylate and lactone forms of the camptothecin derivative irinotecan, CPT-11, and its metabolite SN-38 in plasma. J Chromatogr B Biomed Appl 661: 133–141.
Rivory, L. P. & Robert, J. (1995). Identification and kinetics of a beta-glucuronide metabolite of SN-38 in human plasma after administration of the camptothecin derivative irinotecan. Cancer Chemother Pharmacol 36: 176–179.
Rivory, L. P., Bowles, M. R., Robert, J. & Pond, S. M. (1996). Conversion of irinotecan (CPT-11) to its active metabolite, 7-ethyl-10-hydroxycamptothecin (SN-38), by human liver carboxylesterase. Biochem Pharmacol 52: 1103–1111.
Rothenberg, M., Eckardt, J. R., Kuhn, J. G., Burris, H. A. r., Nelson, J., Hilsenbeck, S. G., Rodriguez, G. I., Thurman, A. M., Smith, L. S., Eckhardt, S. G., Weiss, G. R., Elfring, G. L., Rinaldi, D. A., Schaaf, L. J. & Von-Hoff, D. D. (1996). Phase II trial of irinotecan in patients with progressive or rapidly recurrent colorectal cancer. J Clin Oncol 14: 1128–1135.
Rougier, P., Bugat, R., Douillard, J. Y., Culine, S., Suc, E., Brunet, P., Becouarn, Y., Ychou, M., Marty, M., Extra, J. M., Bonneterre, J., Adenis, A., Seitz, J. F., Ganem, G., Namer, M., Conroy, T., Negrier, S., Merrouche, Y., Burki, F., Mousseau, M., Herait, P. & Mahjoubi, M. (1997). A phase II study of CPT-11 (Irinotecan) in the treatment of advanced colorectal cancer in chemotherapy-naive patients and patients treated with 5FU-based chemotherapy. J Clin Oncol 15: 251–260.
Satoh, T. & Hosokawa, M. (1995). Molecular aspects of carboxylesterase isoforms in comparison with other esterases. Toxicol Lett 82/83: 439–455.
Satoh, T., Hosokawa, M., Atsumi, R., Suzuki, W., Hakusui, H. & Nagai, E. (1994). Metabolic activation of CPT-11, 7-ethyl-10-[4-(1-piperidino)-1-piperidino]carbonyloxycamptothecin, a novel antitumor agent, by carboxylesterase. Biol Pharm Bull 17: 662–664.
Slatter, J. G., Su, P., Sams, J. P., Schaaf, L. J. & Wienkers, L. C. (1997). Bioactivation of the anticancer agent CPT-11 to SN-38 by human hepatic microsomal carboxylesterases and the in vitro assessment of potential drug interactions. Drug Metab Disp 25: 1157–1164.
Slichenmyer, W. J., Rowinsky, E. K., Donehower, R. C. & Kaufmann, S. H. (1993). The current status of camptothecin analogues as antitumor agents. J Natl Cancer Inst 85: 271–291.
Smith, P. K., Krohn, R. I., Hermanson, G. T., Mallia, A. K., Gartner, F. H., Provenzano, M. D., Fijimoto, E. K., Goeke, N. M., Olson, B. J. & Klenk, D. C. (1985). Measurement of protein using bicinchoninic acid. Anal Biochem 150: 76–85.
Takano, H., Kohno, K., Matsuo, K., Matsuda, T. & Kuwano, M. (1992). DNA topoisomerase-targeting antitumor agents and drug resistance. Anticancer Drugs 3: 323–330.
Takasuna, K., Hagiwara, T., Hirohashi, M., Kato, M., Nomura, M., Nagai, E., Yokoi, T. & Kamataki, T. (1996). Involvement of β-glucuronidase in intestinal microflora in the intestinal toxicity of the antitumor camptothecin derivative irinotecan hydrochloride (CPT-11) in rats. Cancer Res 56: 3752–3757.
Tanizawa, A., Fujimori, A., Fujimori, Y. & Pommier, Y. (1994). Comparison of topoisomerase I inhibition, DNA damage, and cytotoxicity of camptothecin derivatives presently in clinical trials. J Natl Cancer Inst 86: 836–842.
Tsuji, T., Kaneda, N., Kado, K., Yokokura, T., Yoshimoto, T. & Tsuru, D. (1991). CPT-11 converting enzyme from rat serum: purification and some properties. J Pharmacobiodyn 14: 341–349.
van Ark-Otte, J., Kedde, M. A., van der Vijgh, W. J. F., Dingemans, A-MC, Jansen, W. J. M., Pinedo, H. M., Boven, E. & Giaccone, G. (1998). Determinants of CPT-11 and SN-38 activities in human lung cancer cells. Br J Cancer 77: 2171–2176.
van der Zee, A. G., de Jong, S., Keith, W. N., Hollema, H., Boonstra, H. & de Vries, E. G. (1994). Quantitative and qualitative aspects of topoisomerase I and II alpha and beta in untreated and platinum/cyclophosphamide treated malignant ovarian tumors. Cancer Res 54: 749–755.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Guichard, S., Terret, C., Hennebelle, I. et al. CPT-11 converting carboxylesterase and topoisomerase I activities in tumour and normal colon and liver tissues. Br J Cancer 80, 364–370 (1999). https://doi.org/10.1038/sj.bjc.6690364
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690364
This article is cited by
-
Irinotecan-Induced Toxicity: A Pharmacogenetic Study Beyond UGT1A1
Clinical Pharmacokinetics (2023)
-
Heterogenous chemosensitivity of a panel of organoid lines derived from small cell neuroendocrine carcinoma of the uterine cervix
Human Cell (2021)
-
Safety and Tolerability of Topotecan-Eluting Radiopaque Microspheres for Hepatic Chemoembolization in a Rabbit Preclinical Model
CardioVascular and Interventional Radiology (2020)
-
Individualization of Irinotecan Treatment: A Review of Pharmacokinetics, Pharmacodynamics, and Pharmacogenetics
Clinical Pharmacokinetics (2018)
-
A phase II study of NK012, a polymeric micelle formulation of SN-38, in unresectable, metastatic or recurrent colorectal cancer patients
Cancer Chemotherapy and Pharmacology (2018)