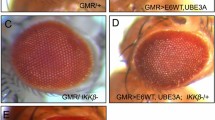
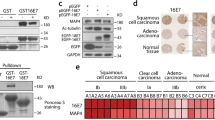
Abstract
Human papillomavirus (HPV) E6 proteins inhibit apoptosis in both p53-dependent and p53-independent manners. A key point in apoptosis is the regulation provided by the Bcl-2 family; and in differentiating keratinocytes, in which HPV replicates, the Bak protein is highly expressed. We show that HPV-18 E6 will inhibit Bak-induced apoptosis and this is mediated by an interaction between the E6 and Bak proteins resulting in degradation of the Bak protein in vivo. We also show that Bak protein interacts with the ubiquitin ligase, E6AP, and that a mutant of Bak defective in E6AP binding is overexpressed in comparison with wild type. These studies suggest that Bak is probably the first naturally occurring target of E6AP to be identified.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
This article is cited by
-
Papillomaviruses and cancer: commonalities and differences in HPV carcinogenesis at different sites of the body
International Journal of Clinical Oncology (2023)
-
Human papillomavirus infection, cervical cancer and the less explored role of trace elements
Biological Trace Element Research (2023)
-
The human papillomavirus E6 protein targets apoptosis-inducing factor (AIF) for degradation
Scientific Reports (2020)
-
A quantitative LumiFluo assay to test inhibitory compounds blocking p53 degradation induced by human papillomavirus oncoprotein E6 in living cells
Scientific Reports (2018)
-
MG132 plus apoptosis antigen-1 (APO-1) antibody cooperate to restore p53 activity inducing autophagy and p53-dependent apoptosis in HPV16 E6-expressing keratinocytes
Apoptosis (2017)