Summary
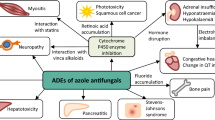
There are 3 main classes of systemic antifungals: the polyene macrolides (e.g. amphotericin B), the azoles (e.g. the imidazoles ketoconazole and miconazole and the triazoles itraconazole and fluconazole) and the allylamines (e.g. terbinafine). Other systemic antifungals include griseofulvin and flucytosine.
Most drug-drug interactions involving systemic antifungals have negative consequences. The interactions of amphotericin B, flucytosine, griseofulvin, terbinafine and azole antifungals can be divided into the following categories: (i) additive dangerous interactions; (ii) modifications of antifungal kinetics by other drugs; and (iii) modifications of the kinetics of other drugs by antifungals.
Amphotericin B and flucytosine mainly interact with other agents pharmacodynamically. Clinically important drug interactions with amphotericin B cause nephrotoxicity, hypokalaemia and blood dyscrasias. The most important drug interaction of flucytosine occurs with myelotoxic agents.
Hypokalaemia can precipitate the long QT syndrome, as well as potentially lethal ventricular arrhythmias like torsade de pointes. Synergism is likely to occur when either QT interval-modifying drugs (e.g. terfenadine and astemizole) and drugs that induce hypokalaemia (e.g. amphotericin B) are coadministered.
Induction and inhibition of cytochrome P450 enzymes at hepatic and extrahepatic sites are the mechanisms that underlie the most serious pharmacokinetic drug interactions of the azole antifungals. These agents have been shown to notably decrease the catabolism of numerous drugs: histamine H1 receptor antagonists, warfarin, cyclosporin, tacrolimus, digoxin, felodipine, lovastatin, midazolam, triazolam, methylprednisolone, glibenclamide (glyburide), phenytoin, rifabutin, ritonavir, saquinavir, nevirapine and nortriptyline. Non-antifungal drugs like carbamazepine, phenobarbital (phenobarbitone), phenytoin and rifampicin (rifampin) can induce the metabolism of azole antifungals. The bioavailability of ketoconazole and itraconazole is also reduced by drugs that increase gastric pH, such as H2 receptor antagonists, proton pump inhibitors, sucralfate and didanosine.
Griseofulvin is an enzymatic inducer of coumarin-like drugs and estrogens, whereas terbinafine seems to have a low potential for drug interactions.
Despite important advances in our understanding of the mechanisms underlying pharmacokinetic drug interactions during the 1990s, at this time they still remain difficult to predict in terms of magnitude in individual patients. This is because of the large interindividual and intraindividual variations in the catalytic activity of those metabolising enzymes that can either be induced or inhibited by various drugs. Notwithstanding these variations, increasing clinical experience is allowing pharmacokinetic interactions to be used to advantage in order to improve the tolerability of some drugs, as recently exemplified by the use of a fixed combination of ketoconazole and cyclosporin.
Similar content being viewed by others
References
Cushard WG, Kohanim M, Lantis LR. Blastomycosis of bone: treatment with intramedullary amphotericin B [abstract]. J Bone Joint Surg 1969; 51A: 704
Miller RP, Bates JH. Amphotericin B toxicity: a follow-up report of 53 patients. Ann Intern Med 1969; 71: 1089–95
Chung DK, Koning MG. Reversible cardiac enlargement during treatment with amphotericin B and hydrocortisone: report of three cases. Am Rev Respir Dis 1971; 103: 831–41
Avery GS. Check-list to potential clinically important interactions. Drugs 1973; 5: 187
Churchill DN, Seely J. Nephrotoxicity associated with combining gentamicin-amphotericin B therapy. Nephron 1977; 19: 176
Gilman AG, Goodman LS, Gilman A, editors. Goodman and Gilmans the pharmacological basis of therapeutics. 6th ed. New York: Macmillan Publishing Co, 1980
Kennedy MS, Deeg HJ, Siegel M, et al. Acute renal toxicity with combined use of amphotericin B and cyclosporine after bone marrow transplantation. Transplantation 1983; 35: 211–5
Stamm AM, Diasio RB, Dismukes WE, et al. Toxicity of amphotericin B plus flucytosine in 194 patients with cryptococcal meningitis. Am J Med 1987; 83: 236–42
Antoniskis D, Larsen RA. Acute, rapidly progressive renal failure with simultaneous use of amphotericin B and pentamidine. Antimicrob Agents Chemother 1990; 34: 470–2
Burger DM, Meenhorst PL, Koks CHW, et al. Drug interactions with zidovudine. AIDS 1993; 113: 445–60
McCauley J. The nephrotoxicity of immunosuppressant agents in modern transplantation. In: Lieberman R, Mukherjee A, editors. Principles of drug development in transplantation & autoimmunity. New York: Chapman & Hall, 1996; 571–8
Hiemenz JW, Walsh TJ. Lipid formulations of amphotericin B: recent progress andfuture directions. Clin Infect Dis 1996; 22 Suppl. 2: S133–44
Quinn DI, Day RO. Drug interactions of clinical importance: an updated guide. Drug Saf 1995; 12: 393–452
Burger DM, Meenhorst PL, Ten Napel CHH, et al. Pharmacokinetic variability of zidovudine in HTV-infected individuals: subgroup analysis and drug interactions. AIDS 1994; 8: 1683–9
Francis P, Walsh JT. Evolving role of flucytosine in immunocompromised patients: new insights into safety, pharmacokinetics, and antifungal therapy. Clin Infect Dis 1992; 15: 1003–18
Harrington JA, Reardon JE, Spector T. 3-Azido-3-deoxythymidine (AZT) monophosphate: an inhibitor of exonucleotic repair of AZT-terminated DNA. Antimicrob Agents Chemother 1993; 37: 918–20
Sales SD, Veal GJ, Hart CA, et al. Zidovudine (ZDV) cytotoxicity in vitro linked to intracellular ZDV-monophosphate concentrations. Br J Pharmacol 1996; 42 Suppl.: 671P–2P
Back DJ, Tjia JF, Abel SM. Azoles, alkylamines and drug metabolism. Br J Dermatol 1991; 126 Suppl. 39: 14–8
Kolars JC, Schmiedlin-Ren P, Schuetz JD, et al. Identification of rifampicin-inducible P450IIIA4 (cyp3A4) in human small bowel enterocytes. J Clin Invest 1992; 90: 1871–8
Watkins PB. Drug metabolism by cytochromes P450 in the liver and small bowel. Gastrointest Pharmacol 1992; 21: 511–26
Gomez DY, Wacher VJ, Tomlanovitch SJ, et al. The effects of ketoconazole on the intestinal metabolism and bioavailability of cyclosporine. Clin Pharmacol Ther 1995; 58: 15–9
Lampen A, Christians U, Bader A, et al. Drug interactions and interindividual variability of cyclosporin metabolism in the small intestine. Pharmacology 1996; 52: 159–68
Nebert DW, Nelson DR, Coon MJ, et al. The P450 superfamily: update on new sequences, gene mapping, and recommended nomenclature. DNA Cell Biol 1991; 10: 1–14
Breckenridge A. Clinical significance of interactions with antifungal agents. Br J Dermatol 1992; 126 Suppl. 39: 19–22
Park BK, Kitteringham NR, Pirmohamed M, et al. Relevance of induction of human drug-metabolizing enzymes: pharmacological and toxicological implications. Br J Pharmacol 1996; 41: 477–91
Guengerich FP. Characterization of human microsomal cytochrome P-450 enzymes. Annu Rev Pharmacol Toxicol 1989; 29: 241–64
Pichard L, Fabre I, Fabre G, et al. Screening for inducers and inhibitors of cytochrome P-450 (cyclosporin A oxydase) in primary cultures of human hepatocytes and liver microsomes. Drug Metab Dispos 1990; 18: 595–606
Maurice M, Pichard L, Daujat M, et al. Effects of imidazole derivatives on cytochromes P-450 from human hepatocytes in primary culture. FASEB J 1992; 6: 752–8
Back DJ, Tjia JF. Comparative effects of the antimycotic drugs ketoconazole, fluconazole, itraconazole and terbinafine on the metabolism of cyclosporin by human liver microsomes. Br J Clin Pharmacol 1991; 32: 624–6
Grant SM, Clissold SP. Fluconazole: a review of its pharmacodynamic and pharmacokinetic properties and therapeutic potential in superficial and systemic mycoses. Drugs 1990; 39: 877–196
Humphrey MJ, Jevons S, Tarbit MH. Pharmacokinetic evaluation of UK-49-858, a metabolically stable triazole antifungal drug, in animals and humans. Antimicrob Agents Chemother 1985; 28: 648–53
Newton DJ, Wang RW, Lu AYH. Cytochrome P450 inhibitors: evaluation of specificities in the in vitro metabolism of therapeutic agents by human liver microsomes. Drug Metab Dispos 1995; 23: 154–8
Sanders G, Dockhorn RJ, Alderman JL, et al. Cardiac effects of acrivastine compared to terfenadine [letter]. J Allergy Clin Immunol 1992; 9: 183
Woosley RL, Chen T, Freiman JP, et al. Mechanism of the cardiotoxic actions of terfenadine. JAMA 1993; 29: 202–9
Woosley RL. Cardiac actions of antihistamines. Annu Rev Pharmacol Toxicol 1996; 36: 233–52
Crane JK, Shik HT. Syncope and cardiac arrhythmia due to an interaction between itraconazole and terfenadine. Am J Med 1993; 95: 445–6
Honing PK, Wortham DC, Zamani K, et al. Terfenadine-ketoconazole interaction. JAMA 1993; 269: 1513–8
Honig PK, Wortham DC, Zamani K, et al. The effects of fluconazole on the steady-state pharmacokinetics and electrocardiographic pharmacodynamics of terfenadine in humans. Clin Pharmacol Ther 1993; 53: 630–6
Pohjola-Sintonen S, Viitasalo M, Toivonen L, et al. Itraconazole prevents terfenadine metabolism and increases risk of torsades de pointes ventricular tachycardia. Eur J Clin Pharmacol 1993; 45: 191–3
Jurima-Romet M, Crawford K, Cyr T, et al. Terfenadine metabolism in human liver: in vitro inhibition by macrolide antibiotics and azole antifungals. Drug Metab Dispos 1994; 22: 849–57
Kivistö KT, Neuvonen PJ, Kotz U. Inhibition of terfenadine metabolism: pharmacokinetic and pharmacodynamic consequences. Clin Pharmacol 1994; 27: 1–6
Lefèbvre RA, Van Peer A, Woestenborghs R. Influence of itraconazole on the pharmacokinetics and electrocardiographic effects of astemizole. Br J Pharmacol 1997; 43: 319–22
Sale ME, Barbey JT, Woosley RL, et al. Electrocardiographic effects of cetirizine in normal subjects. Clin Pharmacol Ther 1994; 56: 295–301
Lippert C, Ling J, Brown P, et al. Mass balance and pharmacokinetics of MDL 16,455A in healthy, male volunteers. Pharm Res 1995; 12: S–390
Simons FE, Bergman JN, Watson TA, et al. The clinical pharmacology of fexofenadine in children. J Allergy Clin Immunol 1996; 98: 1062–4
Fowler NO, McCall D, Chou TC, et al. Electrocardiographic changes and cardiac arrhythmias in patients receiving psychotropic drugs. Am J Cardiol 1976; 37: 223–30
Lathers CM, Lipska LJ. Cardiac arrhythmias, sudden death and psychoactive agents. J Clin Pharmacol 1987; 27: 1–14
Guy JM, André-Fouet X, Porte J, et al. Torsades de pointe et allongement de l’intervalle QT aprés injection de droperidol. Ann Cardiol Angiol 1991; 40: 541–5
Kemper AJ, Dunlap R, Pietro DA. Thioridazine-induced torsades de pointe: successful therapy with isoproterenol. JAMA 1983; 249: 2931–4
Kriwiski M, Perry GY, Tarchisky D, et al. Haloperidol-induced torsades de pointe. Chest 1990; 98: 482–4
Lande G, Drouin E, Gauthier C, et al. Effets arythmogènes du chlorhydrate de sultopride: corrélation clinique et électrophysiologic cellulaire. Ann Fr Anesth Reanim 1992; 11: 629–35
Adamantidis MM, Kerram P, Dupuis BA. In. vitro electrophysiological detection of iatrogenic arrythmogenicity. Fundamental Clin Pharmacol 1994; 8: 391–407
Jansen propulsid concomitant use with nizoral contraindicated. FDC Report — Pink Sheet 1995; Mar 13: T&G14
Lewin MB, Randall MB, Fenrich AL, et al. Cisapride-induced long QT interval. J Pediatr 1996; 128: 279–81
Udall JA. Drug interference with warfarin therapy [letter]. Clin Med 1970; 77: 20
Watson PG, Lochan RG, Redding VJ. Drug interactions with coumarin derivative anticoagulants [letter]. BMJ 1982; 285: 1045–6
Buckley NA, Dawson AH. Drug interactions with warfarin. Med J Aust 1992; 157: 479–83
Smith AG. Potentiation of oral anticoagulants by ketoconazole. BMJ 1984; 288: 188–9
Seaton TL, Celum CL, Black DJ. Possible potentialisation of warfarin by fluconazole. Drug Intell Clin Pharm 1990; 24: 1177–8
Gericke KR. Possible interaction between warfarin and fluconazole. Pharmacotherapy 1993; 13: 508–9
Baciewicz AM, Menke JJ, Bokar JA, et al. Fluconazole-warfarin interaction. Ann Pharmacother 1994; 28: 1111
Colquhoun MC, Daly M, Stewart P, et al. Interaction between warfarin and miconazole oral gel. Lancet 1987; I: 695–6
Back DJ, Stevenson P, Tjia JF. Comparative effects of two antimycotic agents, ketoconazole and terbinafine on the metabolism of tolbutamide, ethinyloestradiol, cyclosporin and ethyoxycoumarin by human liver microsomes in vitro. Br J Clin Pharmacol 1989; 28: 166–70
First MR, Schoeder TJ, Weiskittel P, et al. Concomitant administration of cyclosporin and ketoconazole in renal transplant recipients. Lancet 1989; II: 1198–201
Sugar AM, Saunders C, Idelson BA, et al. Interaction of fluconazole and cyclosporine [letter]. Ann Intern Med 1989; 110: 844
Butman SM, Wild JC, Nolan PE, et al. Prospective study of the safety and financial benefit of ketoconazole as adjunctive therapy to cyclosporine after transplantation. J Heart Lung Transplant 1991; 10: 331–58
Albengres E, Tillement JP. Cyclosporin and ketoconazole drug interaction or therapeutic association? Int J Clin Pharmacol Ther Toxicol 1992; 30: 555–70
Horton CM, Freeman CD, Nolan PE, et al. Cyclosporine interactions with miconazole and other azole-antimycotics: a case report and review of the literature. J Heart Lung Transplant 1992; 11: 1127–32
First RM, Schroeder TJ, Michael A, et al. Cyclosporine-ketoconazole interaction; long term follow-up and preliminary results of a randomized trial. Transplantation 1993; 55: 1000–4
Campana C, Regazzi MB, Buggia I, et al. Clinically significant interactions with cyclosporin: an update. Clin Pharmacokinet 1996; 30: 141–79
Keogh A, Spratt P, McCosker C, et al. Ketoconazole to reduce the need for cyclosporine after cardiac transplantation. N Engl J Med 1995; 333: 628–33
Uwe C, Schmidt G, Bader A, et al. Identification of drugs inhibiting the in vitro metabolism of tacrolimus by human liver microsomes. Br J Clin Pharmacol 1996; 41: 187–90
Osowski CL, Dix SP, Lin LS, et al. Evaluation of the drug interaction between intravenous high-dose fluconazole and cyclosporine or tacrolimus in bone marrow transplant patients. Transplantation 1996; 61: 1268–72
Alderman CP, Jersmann HPA. Digoxin-itraconazole interaction. Med J Aust 1993; 159: 838–9
Sachs MK, Blanchard LM, Green PJ. Interaction of itraconazole and digoxin. Clin Infect Dis 1993; 16: 400–3
McClean KL, Sheehan GT: Interaction between itraconazole and digoxin [letter]. Clin Infect Dis 1994; 18: 259–60
Guenguerich FP. Characterization of human cytochrome P450 enzymes. FASEB J 1992; 6: 745–8
Jalava KM, Olkkola KT, Neuvonen PJ. Itraconazole greatly increases plasma concentrations and effects of felodipine. Clin Pharmacol Ther 1997; 61: 410–5
Neuvonen PJ, Suhonen R. Itraconazole interacts with felodipine. J Am Acad Dermatol 1995; 33: 134–5
Tailor SAN, Gupta AK, Walker SE, et al. Peripheral edema due to nifedipine-itraconazole interaction: a case report. Arch Dermatol 1996; 132: 350–2
Neuvonen PJ, Jalava KM. Itraconazole drastically increases plasma concentrations of lovastatin and lovastatin acid. Clin Pharmacol Ther 1996; 60: 203–8
Lees RS, Lees AM. Rhabdomyolysis from the co-administration of lovastatin and the antifungal agent itraconazole. N Engl J Med 1995; 333: 664–5
Vahre A, Olkkola KT, Neuvonen PJ. Oral triazolam is potentially hazardous to patients receiving systemic antimycotics ketoconazole or itraconazole. Clin Pharmacol Ther 1994; 56: 601–7
Vahre A, Okkola KT, Neuvonen PJ. Fluconazole, but not terbinafine, enhances the effects of triazolam by inhibiting its metabolism. Br J Clin Pharmacol 1996; 41: 319–23
Olkkola KT, Ahonen J, Neuvonen PJ. The effect of systemic antimycotics itraconazole and fluconazole on the pharmacodynamics of intravenous and oral midazolam. Anesth Analg 1996; 82: 511–6
Ahonen J, Olkkola KT, Neuvonen PJ. Effect of the route of administration of fluconazole on the interaction between fluconazole and midazolam. Eur J Clin Pharmacol 1997; 51: 415–9
Paine MF, Shen DD, Kunze KL, et al. First-pass metabolism of midazolam by the human intestine. Clin Pharmacol Ther 1996; 60: 14–24
Loose DS, Kan PB, Hist MA, et al. Ketoconazole blocks adrenal steroidogenesis by inhibiting cytochrome P450-dependent enzymes. J Clin Invest 1983; 71: 1495–9
Pont A, Graybill JR, Craven P, et al. High-dose ketoconazole therapy and adrenal and testicular function in humans. Arch Intern Med 1984; 144: 2150–3
Glynn AM, Slaughter RL, Brass C, et al. Effects of ketoconazole on methylprednisolone pharmacokinetics and cortisol secretion. Clin Pharmacol Ther 1986; 39: 654–9
Kandrotas RJ, Slaughter RL, Brass C, et al. Ketoconazole effects on methylprednisolone disposition and their joint suppression of endogenous Cortisol. Clin Pharmacol Ther 1987; 42: 465–70
Ludwig EA, Slaughter RL, Savliwala M, et al. Steroid-specific effects of ketoconazole on corticosteroid disposition: unaltered prednisolone elimination. Drug Intell Clin Pharm 1989; 23: 858–61
Meurice JC, Lecomte P, Renard JP, et al. Interactions miconazole et sulfamides hypoglycémiants [letter]. Presse Med 1983; 12: 1670
Lazar JD, Wilner KD. Drug interactions with fluconazole. Rev Infect Dis 1990; 12 Suppl. 3: S327–33
Row BR, Thorpe J, Barnett A. Safety of fluconazole in women taking oral hypoglycaemic agents. Lancet 1992; 339: 255–6
Krishnaiah YSR, Satyanarayana S, Visweswaram D. Interaction between tolbutamide and ketoconazole in healthy subjects. Br J Clin Pharmacol 1994; 37: 205–7
Rolan PE, Somogyi AA, Drew MJ, et al. Phenytoin intoxication during treatment with parenteral miconazole [letter]. BMJ 1983; 287: 1760
Howit KM, Oziemski MA. Phenytoin toxicity induced by fluconazole. Med J Aust 1989; 151: 603–4
Mitchell AS, Holland JT. Fluconazole and phenytoin: a predictable interaction [letter]. BMJ 1989; 298: 1315
Blum RA, Wilton JH, Hilligoss DM, et al. Effect of fluconazole on the disposition of phenytoin. Clin Pharmacol Ther 1991; 49: 420–5
Touchette MA, Chandrasekar PH, Milad MA, et al. Contrasting effects of ketoconazole and fluconazole on phenytoin and testosterone disposition in man. Br J Clin Pharmacol 1992; 34: 75–8
Cadle RM, Zenon GJ, Rodriguez-Barradas MC, et al. Fluconazole-induced symptomatic phenytoin toxicity. Ann Pharmacother 1994; 28: 191–5
Fuller JD, Stanfield LED, Craven DE. Rifabutin prophylaxis and Uveitis. N Engl J Med 1994; 330: 1315–6
Narang PK, Trapnell CB, Schoenfelder JR, et al. Fluconazole and enhanced effect of rifabutin prophylaxis. N Engl J Med 1994; 330: 1316–7
Trapnell CB, Narang PK, Li R, et al. Increased plasma rifabutin levels with concomitant therapy in HIV-infected patients. Ann Intern Med 1996; 124: 573–6
Piscitelli SG, Flexner C, Minor JR, et al. Drug interactions in patients infected with human immunodeficiency virus. Clin Infect Dis 1996; 23: 685–93
Kumar GN, Rodrigues D, Buko AM, et al. Cytochrome P450-mediated metabolism of HIV-1 protease inhibitor ritonavir (ABT-538) in liver microsomes. J Pharmacol Exp Ther 1996; 277: 423–31
Murphy RL, Montaner J. Nevirapine: a review of its development, pharmacological profile and potential for clinical use. Exp Opin Invest Drugs 1996; 5: 1183–99
Sahai J, Gallicano K, Pakuts A, et al. Effect of fluconazole on zidovudine pharmacokinetics in patients infected with human immunodeficiency virus. J Infect Dis 1994; 169: 1103–7
Gannon RH, Anderson ML. Fluconazole-nortriptyline drug interaction. Ann Pharmacother 1992; 26: 1456–7
Spina E, Avenoso A, Campo GM, et al. Effect of ketoconazole on the pharmacokinetics of imipramine and desipramine in healthy subjects. Br J Pharmacol 1997; 43: 315–8
Brass C, Galgiani JN, Blaschke TF, et al. Disposition of ketoconazole, an oral antifungal, in humans. Antimicrob Agents Chemother 1982; 21: 151–8
Engelhard D, Stutman HR, Marks MI, et al. Interaction of ketoconazole with rifampin and isoniazid. N Engl J Med 1984; 311: 1681–3
Daneshmend TK, Warnock DW. Clinical pharmacokinetics of systemic antifungal drugs. Clin Pharmacokinet 1988; 14: 13–34
Coker RJ, Tomlinson DR, Parkin J, et al. Interaction between fluconazole and rifampicin [letter]. BMJ 1990; 301: 818
Apseloff G, Hillgoss DM, Gardner MJ, et al. Induction of fluconazole metabolism by rifampicin: in vivo study in humans. J Clin Pharmacol 1991; 31: 358–61
Tucker RM, Denning DW, Hanson LH, et al. Interaction of azoles with rifampicin, phenytoin, and carbamazepine: in vitro and clinical observations. Clin Infect Dis 1992; 14: 165–74
Bonay M, Jonville-Bera AP, Diot P, et al. Possible interaction between phenobarbital, carbamazepine and itraconazole. Drug Saf 1993; 9: 309–11
Drayton J, Dickinson G, Rinaldi MG. Coadministration of rifampicin and itraconazole leads to undetectable levels of serum itraconazole. Clin Infect Dis 1994; 18: 266
Abadie-Kemmerly S, Pankey GA, Dalvision JR. Failure of ketoconazole treatment of blasyomyces dermatidis due to interaction of isoniazid and rifampin [letter]. Ann Intern Med 1988; 109: 844–5
Pilheu JA, Galati MR, Yunis AS, et al. Interaction farmacinetica entre ketoconazole, isoniacida y rifampicina. Medicina 1989; 94: 43–7
Van der Meer JWM, Keuning JJ, Scheijground HW, et al. The influence of gastric acidity on the bioavailability of ketoconazole. J Antimicrob Chemother 1980; 6: 552–4
Lelawongs P, Barone JA, Colaizzi JL, et al. Effect of food and gastric acidity on absorption of orally administered ketoconazole. Clin Pharm 1988; 7: 228–35
Thorpe JE, Baker N, Bromet-Petit M. Effect of oral antacid administration on the pharmacokinetics of oral fluconazole. Antimicrob Agents Chemother 1990; 34: 2032–3
Piscitelli SC, Goss TF, Wilton JH, et al. Effects of ranitidine and sucralfate on ketoconazole bioavailability. Antimicrob Agents Chemother 1991; 35: 1765–71
Goss TF. Evaluation of ketoconazole bioavailability interactions with sucralfate and ranitidine using gastric pH monitoring [abstract]. Clin Pharmacol Ther 1991; 49: 128
Moreno F, Hardin TC, Rinaldi M, et al. Itraconazole-didanosine excipient interaction [letter]. JAMA 1993; 269: 1508
May DB, Drew RH, Yedinak KC, et al. Effect of simultaneous didanosine administration on itraconazole absorption in healthy volunteers. Pharmacotherapy 1994; 14: 509–13
Hoeschele JD, Roy AK, Pecoraro VL, et al. In vitro analysis of the interaction between sucralfate and ketoconazole. Antimicrob Agents Chemother 1994; 38: 319–25
Okkino K, Weibert RT. Warfarin-griseofulvin interaction. Drug Intell Clin Pharm 1986; 20: 291–3
Cote J. Interaction of griseofulvin and oral contraceptives. J Am Acad Dermatol 1990; 22: 124–5
Jensen JC. Pharmacokinetics of Lamisil® in humans. J Dermatol Treat 1990; 1 Suppl. 2: 15–8
Schuster I. The interaction of representative members from two classes of two antimycotics — azoles and allylamines — with cytochromes P-450 in steroidogenic tissues and liver. Xenobiotica 1985; 32: 529–46
Lown KS, Kolars J, Thummel E, et al. Interpatient heterogeneity in expression of CYP3A4 and CYP3A5 in small bowel: lack of prediction by the Erythromycin Breath Test. Drug Metab Dispos 1994; 22: 947–55
Neuvonen PJ, Varhe A, Olkkola T. The effect of ingestion time interval on the interaction between itraconazole and triazolam. Clin Pharmacol Ther 1996; 60: 326–31
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Albengres, E., Le Louët, H. & Tillement, JP. Systemic Antifungal Agents. Drug-Safety 18, 83–97 (1998). https://doi.org/10.2165/00002018-199818020-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002018-199818020-00001