Abstract
Metformin, a biguanide, has been available in the US for the treatment of type 2 diabetes mellitus for nearly 8 years. Over this period of time, it has become the most widely prescribed antihyperglycaemic agent. Its mechanism of action involves the suppression of endogenous glucose production, primarily by the liver. Whether the drug actually has an insulin sensitising effect in peripheral tissues, such as muscle and fat, remains somewhat controversial. Nonetheless, because insulin levels decline with metformin use, it has been termed an ‘insulin sensitiser’. Metformin has also been shown to have several beneficial effects on cardiovascular risk factors and it is the only oral antihyperglycaemic agent thus far associated with decreased macrovascular outcomes in patients with diabetes. Cardiovascular disease, impaired glucose tolerance and the polycystic ovary syndrome are now recognised as complications of the insulin resistance syndrome, and there is growing interest in the management of this extraordinarily common metabolic disorder. While diet and exercise remain the cornerstone of therapy for insulin resistance, pharmacological intervention is becoming an increasingly viable option. We review the role of metformin in the treatment of patients with type 2 diabetes and describe the additional benefits it provides over and above its effect on glucose levels alone. We also discuss its potential role for a variety of insulin resistant and prediabetic states, including impaired glucose tolerance, obesity, polycystic ovary syndrome and the metabolic abnormalities associated with HIV disease.



Similar content being viewed by others
References
Witters LA. The blooming of the French lilac. J Clin Invest 2001; 108: 1105–7
Kolata GB. The phenformin ban: is the drug an imminent hazard? Science 1979; 203: 1094–6
Bailey CJ, Turner RC. Metformin. N Engl J Med 1996; 334: 574–9
Inzucchi SE, Maggs DG, Spollett GR, et al. Efficacy and metabolic effects of metformin and troglitazone in type II diabetes mellitus. N Engl J Med 1998; 338: 867–72
Inzucchi SE. Metformin or thiazolidinediones as first-line therapy for type 2 diabetes: focus on cardiovascular protection. Pract Diabetol 2002; 21: 7–12
Stadtmauer LA, Wong BC, Oehninger S. Should patients with polycystic ovary syndrome be treated with metformin?: benefits of insulin sensitizing drugs in polycystic ovary syndrome: beyond ovulation induction. Hum Reprod 2002; 17(12): 3016–26
Cohn G, Valdes G, Capuzzi DM. Pathophysiology and treatment of the dyslipidemia of insulin resistance. Curr Cardiol Rep 2001; 3(5): 416–23
Mehnert H. Metformin, the rebirth of a biguanide: mechanism of action and place in the prevention and treatment of insulin resistance. Exp Clin Endocrinol Diabetes 2001; 109 Suppl. 2: S259–64
DeFronzo RA. The triumvirate: β-cell, muscle, liver: a collusion responsible for NIDDM. Diabetes 1988; 37: 667–87
Jeng CY, Shen WH, Fuh MM, et al. Relationship between hepatic glucose production and fasting glucose concentration in patients with NIDDM. Diabetes 1994; 43: 1440–4
Hundal RS, Krssak M, Dufour S, et al. Mechanism by which metformin reduces glucose production in type 2 diabetes. Diabetes 2000; 49: 2063–9
Magnusson I, Rothman DL, Katz LD, et al. Increased rate of gluconeogenesis in type II diabetes mellitus. J Clin Invest 1992; 90: 1323–7
Cusi K, DeFronzo RA. Metformin: a review of its metabolic effects. Diabetes Rev 1998; 6: 89–131
Garber AJ, Duncan TG, Goodman AM, et al. Efficacy of metformin in type II diabetes: results of a double-blind, placebo-controlled, dose-response trial. Am J Med 1997; 102: 491–7
DeFronzo RA, Goodman AM, for the Multicenter Metformin Study Group. Efficacy of metformin in NIDDM patients poorly controlled on diet alone or diet plus sulfonylurea. N Engl J Med 1995; 333: 541–9
Stumvoll M, Nurjhan N, Periello G, et al. Metabolic effects of metformin in non-insulin dependent diabetes mellitus. N Engl J Med 1995; 333: 550–4
Cusi K, Consoli A, DeFronzo RA. Metabolic effects of metformin on glucose and lactate metabolism in non insulin-dependent diabetes mellitus. J Clin Endocrinol Metab 1996; 81: 4059–67
Christiansen MP, Linfoot PA, Neese RA, et al. Metformin: effects upon postabsorptive intrahepatic carbohydrate fluxes [abstract]. Diabetes 1997; 46 Suppl. 1: 244A
Yu JG, Kruszynska YT, Mulford MI, et al. A comparison of troglitazone and metformin on insulin requirements in euglycemic intensively insulin-treated type 2 diabetic patients. Diabetes 1999; 48: 2414–21
Radziuk J, Zhang Z, Wiernperger N, et al. Effects of metformin on lactate uptake and gluconeogenesis in the perfused rat liver. Diabetes 1997; 46: 1406–13
Argaud D, Roth H, Wiernsperger N, et al. Metformin decreases gluconeogenesis by enhancing the pyruvate kinase flux in isolated rat hepatocytes. Eur J Biochem 1993; 213: 1341–8
Large V, Beylot M. Modifications of citric acid cycle activity and gluconeogenesis in streptozocin-induced diabetes and effects of metformin. Diabetes 1999; 48: 1251–7
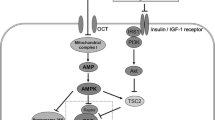
Zhou G, Myers R, Li Y, et al. Role of AMP-activated protein kinase in the mechanism of metformin action. J Clin Invest 2001; 108: 1167–74
Owen MR, Doran E, Halestrap AP, et al. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem J 2000; 348: 607–14
Hawley SA, Gadalla AE, Olsen GS, et al. The antidiabetic drug metformin activates the AMP-activated protein kinase cascade via an adenine nucleoside-independent mechanism. Diabetes 2002; 51: 2420–5
Lochhead PA, Salt IP, Walker KS, et al. 5-aminoimidazole-4-carboxamide riboside mimics the effects of insulin on the expression of the 2 key gluconeogenic genes PEPCK and glucose-6-phosphatase. Diabetes 2009; 49: 896–903
Bergeron R, Russell RR, Young LH, et al. Effects of AMPK activation on muscle glucose metabolism in conscious rats. Am J Physiol 1999; 276: E938–44
Dominguez LJ, Davidoff AJ, Srinivas PR, et al. Effects of metformin on tyrosine kinase activity, glucose transport, and intracellular calcium in rat vascular smooth muscle. Endocrinology 1996; 137: 113–21
Yki-Jarvinen H, Nikkila K, Makimattila S. Metformin prevents weight gain by reducing dietary intake during insulin therapy in patients with type 2 diabetes mellitus. Drugs 1999; 58 Suppl. 1: 53–4
Davidoff F, Bertolini D, Haas D. Enhancement of the mitochondrial Ca2+ uptake rate by phenethylbiguanide and other organic cations with hypoglycemic activity. Diabetes 1978; 27: 757–65
Wiernsperger NF, Bailey CJ. The antihyperglycemic effect of metformin: therapeutic and cellular mechanisms. Drugs 1999; 58 Suppl. 1: 31–9
Hundal HS, Ramlal T, Reyes R, et al. Cellular mechanism of metformin action involves glucose transporter translocation from an intracellular pool to the plasma membrane in L6 muscle cell. Endocrinology 1992; 131: 1165–73
Stith BJ, Goalstone ML, Espinoza R, et al. The antidiabetic drug metformin elevates receptor tyrosine kinase activity and inositol 1,4,5-trisphosphate mass in Xenopus oocytes. Endocrinology 1996; 137: 2990–9
DeFronzo RA, Barzilai N, Siminson DC. Mechanism of metformin action in obese and lean non-insulin dependent diabetic subjects. J Clin Endocrinol Metab 1991; 73: 1294–301
Perriello G, Misericordia P, Volpi E, et al. Acute antihyperglycemic mechanisms of metformin in NIDDM: evidence for suppression of lipid oxidation and hepatic glucose production. Diabetes 1994; 43: 920–8
Sindelar DK, Chu CA, Rohlie M, et al. The role of fatty acids in mediating the effects of peripheral insulin on hepatic glucose production in the conscious dog. Diabetes 1997; 46: 187–96
Bergman RN, Mittleman SD. Central role of the adipocyte in insulin resistance. J Basic Clin Physiol Pharmacol 1998; 9: 205–21
Dresner A, Laurent D, Marcucci M, et al. Effects of free fatty acids on glucose transport and IRS-1-associated phosphatidylinositol 3-kinase activity. J Clin Invest 1999; 103: 253–9
Patane G, Piro S, Rabuazzo AM, et al. Metformin restores insulin secretion altered by chronic exposure to free fatty acids or high glucose: a direct metformin effect on pancreatic beta-cells. Diabetes 2000; 49: 735–40
Wilcock C, Bailey CJ. Accumulation of metformin by tissues of the normal and diabetic mouse. Xenobiotica 1999; 24: 49–57
Ikeda T, Iwata K, Murakami H. Inhibitory effect of metformin on intestinal glucose absorption in the perfused rat intestine. Biochem Pharmacol 2000; 59: 887–90
Scheeen AJ. Clinical pharmacokinetics of metformin. Clin Pharmacokinet 1996; 30: 359–71
DeFronzo RA. Pharmacologic therapy for type 2 diabetes mellitus. Ann Intern Med 1999; 131: 281–303
Misbin RI, Green L, Stadel BV, et al. Lactic acidosis in patients with diabetes treated with metformin. N Engl J Med 1998; 338: 265–6
Lalau JD, Race JM. Prognostic value of arterial lactate levels and plasma metformin concentrations. Drug Saf 1999; 20: 377–84
Bailey CJ, Wilcock C, Day C. Effect of metformin on glucosemetabolism in the splanchic bed. Br J Pharmacol 1992; 105: 1009–13
Hermann LS, Schersten B, Bitzen PO, et al. Therapeutic comparisons of metformin and sulfonylurea, alone and in various combinations: a double blind controlled study. Diabetes Care 1994; 17: 1100–9
United Kingdom Prospective Diabetes Study (UKPDS) Group. Relative efficacy of randomly allocated diet, sulfonylurea, insulin, or metformin in patients with newly diagnosed non-insulin dependent diabetes followed for three years (UKPDS 13). BMJ 1995; 310: 83–8
Johnson K. Efficacy of metformin in the treatment of NIDDM. Meta-analysis. Diabetes Care 1999; 22: 33–7
United Kingdom Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998; 352: 854–65
Kirpichnikov D, McFarlane SI, Sowers JR. Metformin: an update. Ann Intern Med 2002; 137: 25–33
Inzucchi SI. Oral antihyperglycemic therapy for type 2 diabetes. JAMA 2002; 287: 360–72
Dornan T, Heller S, Peck G, et al. Double blind evaluation of efficacy and tolerability of metformin in NIDDM. Diabetes Care 1991; 14: 342–4
Fontbonne A, Charles MA, Juhan-Vague I, et al. The effect of metformin on the metabolic abnormalities associated with upper-body fat distribution: BIGPRO Study Group. Diabetes Care 1996; 19(9): 920–6
Glueck CJ, Wang P, Fontaine R, et al. Metformin induced resumption of normal menses in 39 of 43 (91%) previously amenorrheic women with the polycystic ovary syndrome. Metabolism 1999; 48: 511–9
Pasquali R, Gambineri A, Biscotti D, et al. Effect of long term treatment with metformin added to hypocaloric diet on body composition, fat distribution, and androgen and insulin levels in abdominally obese women with and without the polycystic ovary syndrome. J Clin Endocrinol Metab 2000; 85: 2161–74
Turner RC, Cull CA, Frighi V, et al. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirements for multiple therapies (UKPDS 49). UK Prospective Diabetes Study (UKPDS) Group. JAMA 1999; 281: 2005–12
Haupt E, Knick B, Koschinsky T, et al. Oral antidiabetic combination therapy with sulphonylurea and metformin. Diabetes Metab 1991; 17: 224–31
Reaven GM, Johnston P, Hollenbeck CB, et al. Combined metformin-sulfonylurea treatment of patients with non-insulin dependent diabetes in fair to poor glycemic control. J Clin Endocrinol Metab 1992; 74: 1020–6
Riddle M. Combining sulfonylureas and other oral agents. Am J Med 2000; 108 Suppl. 6a: 15S–22S
Jaber LA, Nowak SN, Slaughter RR. Insulin-metformin combination therapy in obese patients with type 2 diabetes. J Clin Pharmacol 2002; 42: 89–94
Hermann LS, Kalen J, Katzman P, et al. Long term glycemic improvement after addition of metformin to insulin in insulin treated obese type 2 diabetes patients. Diabetes Obes Metab 2001; 3: 428–34
Bailey CJ. Metformin: a useful adjunct to insulin therapy? Diabet Med 2000; 17: 83–4
Rosenstock J, Brown A, Fischer J, et al. Efficacy and safety of acarbose in metformin treated patients with type 2 diabetes. Diabetes Care 1998; 21: 2050–5
Chiasson JL, Naditch L. Canadian University Investigator Group. The synergistic effect of miglitol plus metformin combination therapy in the treatment of type 2 diabetes. Diabetes Care 2001; 24: 989–94
Einhorn D, Rendell M, Rosenzweig J, et al. Pioglitazone hydrochloride in combination with metformin in the treatment of type 2 diabetes mellitus: a randomized placebo-controlled study. The Pioglitazone 027 Study Group. Clin Ther 2000; 22: 1395–409
Fonesca V, Rosenstock J, Patwardhan R, et al. Effect of metformin and rosiglitazone combination therapy in patients with type 2 diabetes mellitus: a randomized controlled trial. JAMA 2000; 283: 1695–702
Moses R. Repaglinide in combination therapy with metformin in type 2 diabetes. Exp Clin Endocrinol Diabetes 1999; 107 Suppl. 4: 136–9
Moses R, Slobodunik R, Boyages S, et al. Effect of repaglinide addition to metformin monotherapy on glycemic control in patients with type 2 diabetes. Diabetes Care 1999; 22: 119–24
Marre M, Van Gaal L, Usadel KH, et al. Nateglinide improves glycemic control when added to metformin monotherapy: results of a randomized trial with type 2 diabetes patients. Diabetes Obes Metab 2002; 4: 177–86
Horton ES, Clinkingbeard C, Gatlin M, et al. Nateglinide alone and in combination with metformin improves glycemic control by reducing meal time glucose levels in type 2 diabetes. Diabetes Care 2000; 23: 1660–5
Hirschberg Y, Karara AH, Pietri AO, et al. Improved control of mealtime glucose excursions with coadministration of nateglinide and metformin. Diabetes Care 2000; 23: 349–53
Pagano G, Tagliaferro V, Carta Q, et al. Metformin reduces insulin requirement in type 1 (insulin-dependent) diabetes. Diabetologia 1983; 24: 351–4
Stratton IM, Adler AI, Neil HA, et al. Association of glycemia with microvascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ 2000; 321: 405–12
Nagi DK, Yudkin JS. Effects of metformin on insulin resistance, risk factors for cardiovascular disease, and plasminogen activator inhibitor in NIDDM subjects: a study of two ethnic groups. Diabetes Care 1993; 16: 621–9
Olsson J, Lindberg G, Gottsater M, et al. Increased mortality in type II diabetic patients using sulfonylurea and metformin in combination: a population based observational study. Diabetolgia 2000; 43: 558–60
Despres JP, Lamarche B, Mauriege P, et al. Hyperinsulinemia as an independent risk factor for ischemic heart disease. N Engl J Med 1996; 334: 952–7
Haffner SM. Epidemiology of insulin resistance and its relation to coronary artery disease. Am J Cardiol 1999; 84: 11J-4J
Simplicini A, Del Prato S, Giusto M, et al. Short term effects of metformin on insulin sensitivity and sodium homeostasis in essential hypertensives. J Hypertens 1993; 11 Suppl. 5: S276–7
Robinson AC, Burke J, Robinson S, et al. The effects of metformin on glycemic control and serum lipids in insulin treated NIDDM patients with suboptimal control. Diabetes Care 1998; 21: 701–5
Abasi F, Kamath V, Rizvi AA, et al. Results of a placebo controlled study of the metabolic effects of the addition of metformin to sulfonylurea treated patients: evidence for a central role of adipose tissue. Diabetes Care 1997; 20: 1863–9
Wu MS, Johnston P, Sheu WH, et al. Effect of metformin on carbohydrate and lipoprotein metabolism in NIDDM patients. Diabetes Care 1990; 13: 1–8
Velazquez EM, Mendoza SG, Wang P, et al. Metformin therapy is associated with a decrease in plasma plasminogen activator inhibitor-1, lipoprotein (a), and immunoreactive insulin levels in patients with the polycystic ovary syndrome. Metabolism 1997; 46: 454–7
Bell DS, Ovalle F. Metformin lowers lipoprotein (a) levels. Diabetes Care 1998; 21: 2028
Fontbonne A, Charles MA, Juhan-Vague I, et al. The effect of metformin on the metabolic abnormalities associated with upper body fat distribution: BIGPRO study group. Diabetes Care 1996; 19: 920–6
Grant PJ, Stickland MH, Booth NA, et al. Metformin causes a reduction in basal and post-venous occlusion plasminogen activator inhibitor-1 in type 2 diabetic patients. Diabet Med 1991; 8: 361–5
Charles MA, Morange P, Eschwege E, et al. Effect of weight change and metformin on fibrinolysis and the von Willebrand factor in obese non-diabetic subjects: the BIGPRO study. Biguanides and the prevention of the risk of obesity. Diabetes Care 1998; 21: 1967–72
Marfella R, Acampora R, Verrazzo G, et al. Metformin improves hemodynamic and rheological responses to L-arginine in NIDDM patients. Diabetes Care 1998; 19: 934–9
Landin K, Tengborn L, Smith U. Treating insulin resistance in hypertension with metformin reduces both blood pressure and metabolic risk factors. J Intern Med 1991; 229: 181–7
Desouza C, Keebler M, McNamara DB, et al. Drugs affecting homocysteine metabolism. Drugs 2002; 62: 605–16
Vrbikova J, Bicikova M, Tallova J, et al. Homocysteine and steroid levels in metformin treated women with polycystic ovary syndrome. Exp Clin Endocrinol Diabetes 2002; 110: 74–6
Ferrannini E, Buzzigoli G, Bonadonna R, et al. Insulin resistance in essential hypertension. N Engl J Med 1987; 317: 350–7
Taylor AA. Pathophysiology of hypertension and endothelial dysfunction in patients with diabetes. Endocrinol Metab Clin North Am 2001; 30: 983–97
Giugliano D, Quatraro A, Consoli G, et al. Metformin for obese, insulin treated diabetic patients: improvement in glycemic control and reduction of metabolic risk factors. Eur J Clin Pharmacol 1993; 44: 107–12
Mather KJ, Verma S, Anderson TJ. Improved endothelial function with metformin in type 2 diabetes mellitus. J Am Coll Cardiol 2001; 37: 1344–50
Diamanti-Kandarakis E, Spina G, Kouli C, et al. Increased endothelial-1 levels in women with polycystic ovary syndrome and the beneficial effect of metformin therapy. J Clin Endocrinol Metab 2001; 86: 4666–73
Ridker PM, Glyn RJ, Hennekens CH. C-reactive protein adds to the predictive value of total and HDL cholesterol in determining risk of first myocardial infarction. Circulation 1998; 97: 2007–11
Chu NV, Kong AP, Kim DD, et al. Differential effects of metformin and troglitazone on cardiovascular risk factors in patients with type 2 diabetes. Diabetes Care 2002; 25: 542–9
Ludwig DS, Ebbeling CB. Type 2 diabetes mellitus in children: primary care and public health considerations. JAMA 2001; 286: 1427–30
Fagot-Campagna A, Pettitt DJ, Engelgau MM, et al. Type 2 diabetes among North American children and adolescents: an epidemiologic review and a public health perspective. J Pediatr 2000; 136: 664–72
Freemark M, Bursey D. The effects of metformin on body mass index and glucose tolerance in obese adolescents with fasting hyperinsulinemia and a family history of type 2 diabetes. Pediatrics 2001; 107(4): E55
Lustig R. Childhood obesity. In: A current review of pediatric endocrinology 1999. Rockland (MA): Serono Symposia, 1999: 133–9
Jones KL, Arslanian S, Peterokova VA, et al. Effect of metformin in pediatric patients with type 2 diabetes: a randomized controlled trial. Diabetes Care 2002; 25: 89–94
Morrison JA, Cottingham EM, Barton BA. Metformin for weight loss in pediatric patients taking psychotropic drugs. Am J Psychiatry 2002; 159: 655–7
Pi-Sunyer FX. Medical hazards of obesity. Ann Intern Med 1993; 119: 655–60
Ford ES, Williamson DF, Liu S. Weight change and diabetes incidence: finding from a national cohort of US adults. Am J Epidemiol 1997; 146: 214–22
Tuomilehto J, Lindstrom J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001; 344: 1343–50
Knowler WC, Barrett Connor E, Fowler SE, et al. Reduction inthe incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002; 346(6): 393–403
Futterweit W. Polycystic ovary syndrome: clinical perspectives and management. Obstet Gynecol Surv 1999; 54: 403–13
Dunaif A, Graf M, Mandeli J, et al. Characterization of groups of hyperandrogenemic women with acanthosis nigricans, impaired glucose tolerance, and/or hyperinsulinemia. J Clin Endocrinol Metab 1987; 65: 499–507
Dunaif A, Segal KR, Futterweit W, et al. Profound insulin resistance, independent of obesity, in polycystic ovary syndrome. Diabetes 1989; 38: 1165–74
Nestler JE, Stovall D, Akhter N, et al. Strategies for the use of insulin sensitizing drugs to treat infertility in women with polycystic ovary syndrome. Fertil Steril 2002; 77: 209–15
Pugeat M, Ducluzeau PH. Insulin resistance, polycystic ovary syndrome and metformin. Drugs 1999; 58 Suppl. 1: 41–6
Dunaif A. Insulin resistance and the polycystic ovary syndrome: mechanisms and implications for pathogenesis. Endocr Rev 1997; 18: 774–800
Nestler JE, Powers LP, matt DW, et al. A direct effect of hyperinsulinemia on serum sex hormone-binding globulin levels in obese women with the polycystic ovary syndrome. J Clin Endocrinol Metab 1991; 72: 83–9
Bloomgarden ZT, Futterweit W, Poretsky L. Use of insulin sensitizing agents in patients with polycystic ovary syndrome. Endocr Pract 2001; 7: 279–86
Morin-Papunen LC, Vanhkonen I, Koivunen RM, et al. Endocrine and metabolic effects of metformin versus ethinyl estradiol-cyproterone acetate in obese women with polycystic ovary syndrome: a randomized study. J Clin Endocrinol Metab 2000; 85: 3161–8
Moghetti P, Castello R, Negri C, et al. Metformin effects on clinical features, endocrine and metabolic profiles, and insulin sensitivity in polycystic ovary syndrome: a randomized, double blind, placebo-controlled 6 month trial, followed by open, long term clinical evaluation. J Clin Endocrinol Metab 2000; 85: 139–46
Valazquez E, Acosta A, Mendoza SG. Menstrual cyclicity after metformin treatment in polycystic ovary syndrome. Obstet Gynecol 1997; 90: 392–5
La Marca A, Egbe TO, Morgante G, et al. Metformin treatment reduces ovarian cytochrome p-450c17alpha response to human gonadotropin in women with insulin resistance-related polycystic ovary syndrome. Hum Reprod 2000; 15: 21–3
Kelly CJ, Gordon D. The effects of metformin on hirsuitism in polycystic ovary syndrome. Eur J Endocrinol 2002; 147: 217–21
Velazquez EM, Mendoza S, Hamer T, et al. Metformin therapy in polycystic ovary syndrome reduces hyperinsulinemia, insulin resistance, hyperandrogenism, and systolic blood pressure, while facilitating normal menses and pregnancy. Metabolism 1994; 43: 647–54
Morin-Papunen LC, Koivunen RM, Ruokonen A, et al. Metformin therapy improves the menstrual pattern with minimal endocrine and metabolic effects in women with polycystic ovary syndrome. Fertil Steril 1998; 69: 691–6
Nestler JE, Jakubowicz DJ, Evans WS, et al. Effects of metformin on spontaneous and clomiphene-induced ovulation in the polycystic syndrome. N Engl J Med 1998; 338: 1876–80
Vandermolen DT, Ratts VS, Evans WS, et al. Metformin increases the ovulatory rate and pregnancy rate from clomiphene citrate in patients with polycystic ovary syndrome who are resistant to clomiphene citrate alone. Fertil Steril 2001; 75: 310–5
Acbay O, Gundogdu S. Can metformin reduce insulin resistance in polycystic ovary syndrome? Fertil Steril 1996; 65: 946–9
Ehrmann DA, Cavaghan MK, Imperial J, et al. Effects of metformin on insulin secretion, insulin action, and ovarian steroidogenesis in women with polycystic ovary syndrome. J Clin Endocrinol Metab 1997; 82: 524–30
De Leo V, La Marca A, Ditto A, et al. Effects of metformin on gonadotropin-induced ovulation in women with polycystic ovary syndrome. Fertil Steril 1999; 72: 282–5
Stadtmauer LA, Toma SK, Riehl RM, et al. Metformin treatment of patients with polycystic ovary syndrome undergoing in vitro fertilization improves outcomes and is associated with modulation of the insulin-like growth factors. Fertil Steril 2001; 75(3): 505–9
Glueck CJ, Phillips H, Cameron D, et al. Continuing metformin throughout pregnancy in women in polycystic ovary syndrome appears to safely reduce first trimester spontaneous abortion: a pilot study. Fertil Steril 2001; 75: 46–52
Jakubowicz DJ, Iuorno MJ, Jakubowicz S, et al. Effects of metformin on early pregnancy loss in the polycystic ovary syndrome. J Clin Endocrinol Metab 2002; 87(2): 524–9
Coetzee EJ, Jackson WPU. Metformin in management of pregnant insulin-independent diabetics. Diabetologia 1979; 16: 241–5
Coetzee EJ, Jackson WP. The management of non-insulin dependent diabetes during pregnancy. Diabetes Res Clin Pract 1985; 1: 281–7
Azziz R, Ehramann D, Legro RS, et al. Troglitazone improves ovulation and hirsutism in the polycystic ovary syndrome: a multicenter, double-blind, placebo-controlled trial. J Clin Endocrinol Metab 2001; 86: 1626–32
Dunaif A, Scott D, Finewood D, et al. The insulin-sensitizing agent troglitazone improves metabolic and reproductive abnormalities in the polycystic ovary syndrome. J Clin Endocrinol Metab 1996; 81: 3299–306
Glueck CJ, Wang P, Kobayashi S, et al. Metformin therapy throughout pregnancy reduces the development of gestational diabetes in women with polycystic ovary syndrome. Fertil Steril 2002; 77: 520–5
Carr A, Samaras K, Thorisdottir A, et al. Diagnosis, prediction and natural course of HIV-1 protease-inhibitor-associated lipodystrophy, hyperlipidemia, and diabetes mellitus: a cohort study. Lancet 1999; 353: 2093–9
Hadigan C, Miller K, Corcoran C, et al. Fasting hyperinsulinemia and changes in regional body composition in human immunodeficiency virus-infected women. J Clin Endocrinol Metab 1999; 84: 1932–7
Noor M, Lo J, Mulligan K, et al. Metabolic effects of indinavir in healthy HIV-seronegative men. AIDS 2001; 15: 11–8
Murata H, Hruz PW, Mueckler M. The mechanism of insulin resistance caused by HIV protease inhibitor therapy. J Biol Chem 2000; 275: 20251–4
Hadigan C, Meigs JB, Rabe J, et al. Increased PAI-1 and tPA antigen levels are reduced with metformin therapy in HIV-infected patients with fat redistribution and insulin resistance. J Clin Endocrinol Metab 2001; 86: 939–43
Hadigan C, Corcoran C, Basgoz N, et al. Metformin in the treatment of HIV lipodystrophy syndrome: a randomized controlled trial. JAMA 2000; 284: 472–7
Saint-Marc T, Touraine JL. Effects of metformin on insulin resistance and central adiposity in patients receiving effective protease inhibitor therapy. AIDS 1999; 13: 1000–2
Glueck CJ, Fontaine RN, Wang P, et al. Metformin reduces weight, centripetal obesity, insulin, leptin, and low density lipoprotein cholesterol in nondiabetic, morbidly obese subjects with body mass index greater than 30. Metabolism 2001; 50: 856–61
Kay JP, Alemzadeh R, Langley G, et al. Beneficial effects of metformin in normoglycemic morbidly obese adolescents. Metabolism 2001; 50: 1457–61
Gokel A, Gumurdulu Y, Karakose H, et al. Evaluation of the safety and efficacy of sibutramine, orlistat and metformin in the treatment of obesity. Diabetes Obes Metab 2002; 4: 49–55
Dorella M, Giusto M, Da Tos V, et al. Improvement of insulin sensitivity by metformin treatment does not lower blood pressure if nonobese insulin-resistant hypertensive patients with normal glucose tolerance. J Clin Endocrinol Metab 1996; 81: 1568–74
Charles MA, Eschwege E, Grandmottet P, et al. Treatment with metformin of non-diabetic men with hypertension, hypertriglyceridemia and central fat distribution: the BIGPRO 1.2 trial. Diabetes Metab Res Rev 2000; 16: 2–7
Giugliano D, De Rosa N, Di Maro G, et al. Metformin improves glucose, lipid metabolism, and reduces blood pressure in hypertensive, obese women. Diabetes Care 1993; 16: 1387–90
Mannucci E, Ognibene A, Cremasco F, et al. Effect of metformin on glucagon-like peptide (GLP-1) and leptin levels in obese non-diabetic subjects. Diabetes Care 2001; 24: 489–94
Angulo P. Nonalcoholic fatty liver disease. N Engl J Med 2002; 346: 1221–31
Lin HZ, Yang SQ, Chuckaree C, et al. Metformin reverses fatty liver disease in obese, leptin-deficient mice. Nat Med 2000; 6: 998–1003
Marchesini G, Brizi M, Bianchi G, et al. Metformin in nonalcoholic steatohepatitis. Lancet 2001; 358: 893–4
Acknowledgements
Dr Hundal and Dr Inzucchi have received honoraria from and/or are on speaker’s bureaus for Glaxo-Smith Kline, Takeda Pharmaceuticals North America, and Bristol-Myers Squibb. Dr Inzucchi has also received research grant support from Bristol-Myers Squibb.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hundal, R.S., Inzucchi, S.E. Metformin. Drugs 63, 1879–1894 (2003). https://doi.org/10.2165/00003495-200363180-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-200363180-00001