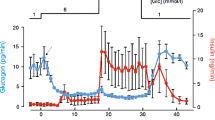
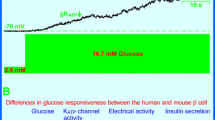
Summary
Insulin secretion by the pancreatic Beta cell is dependent upon transmembrane ion fluxes gated by the ATP-regulated potassium channel and the voltage regulated, L-type calcium channel. This work group examined major recent advances in the structure and modulation of ion channels and how those advances may pertain to the physiology of insulin secretion and the pharmacological treatment of Type 2 (non-insulin-dependent) diabetes mellitus. Structural studies have revealed that voltage gated ion channels are related, complex, and comprised of multiple components: sodium channels consist of three distinct subunits. L-type calcium channels, crucial to the insulin secretory response are structurally related to the sodium channel but contain additional subunits. Potassium channels are less closely related and appear to function as homotetramers. Modulation of ion channel activity is similarly complex: site specific phosphorylation by multiple protein kinases under the control of several intracellular second messenger systems may increase or decrease conductance. Subunit composition and relatively stable changes in the modal state of ion channels also appear to be critical to ion channel gating properties. Functional studies of the Beta-cell ATP-regulated potassium channel suggest two distinct nucleotide binding sites which link this channel to the metabolic state of the Beta cell. The multiple paths of ion channel modulation provide multiple targets for therapeutic intervention. Where detailed characterisation of ion channel structure has been achieved, those targets are being used for specific drug design. Such complete characterisation has not yet been achieved for Beta-cell ion channels and this presents a major goal for diabetes research.
Article PDF
Similar content being viewed by others
References
Catterall WA (1992) Cellular and molecular biology of sodium channels. Physiol Rev (in press)
Isom LL, De Jongh KS, Patton DE et al. (1992) Primary structure and functional expression of the β 1 subunit of the rat brain sodium channel. Science 256:839–842
Numann R, Catterall WA, Scheuer T (1991) Functional modulation of brain sodium channels by protein kinase C phosphorylation. Science 254:115–118
West JW, Numann R, Murphy BJ, Scheuer T, Catterall WA (1991) Identification of a phosphorylation site in a conserved intracellular loop that is required for modulation of sodium channels by protein kinase C. Science 254:866–868
Li M, West JW, Lai Y, Scheuer T, Catterall WA (1992) Functional modulation of brain sodium channels by cAMP-dependent phosphorylation. Neuron 8:1151–1159
Miller C (1991) 1990: Annus mirabilis of potassium channels. Science 252:1092–1096
Hoshi T, Zagotta WN, Aldrich RW (1991) Two types of inactivation in Shaker K+ channels: effects of alterations in the carboxy-terminal region. Neuron 7:547–556
Hoshi T, Zagotta WN, Aldrich RW (1990) Biophysical and molecular mechanisms of Shaker potassium channel inactivation. Science 250:533–538
Zagotta WN, Hoshi T, Aldrich RW (1990) Restoration of inactivation in mutants of Shaker potassium channels by a peptide derived from ShB. Science 250:568–571
Glossmann H, Striessnig J (1990) Molecular properties of calcium channels. Rev Physiol Biochem Pharmacol 114:1–105
Striessnig J, Glossmann H, Catterall WA (1990) Identification of a phenylalkylamine binding region within the α1 subunit of skeletal muscle Ca2+ channels. Proc Natl Acad Sci USA 87:9108–9112
Striessnig J, Catterall WA (1991) The dihydropyridine receptor of L-type Ca2+-channels: identification of binding domains for (+)-[3H]PN200-110 and [3H]azidopine within the α1 subunit. Proc Natl Acad Sci USA 88:10769–10773
Regulla S, Schneider T, Nastainczyk W, Meyer HE, Hofmann F (1991) Identification of the site of interaction of the dihydropyridine channel blockers nitrendipine and azidopine with the calcium-channel α1 subunit. EMBO J 10:45–49
Singer D, Biel M, Lotan I, Flockerzi V, Hofmann F, Dascal N (1991) The roles of the subunits in the function of the calcium channel. Science 253:1553–1557
Bosse E, Bottlender R, Kleppisch T et al. (1992) Stable and functional expression of the calcium channel α1 subunit from smooth muscle in somatic cell lines. EMBO J 11 (in press)
Hullin R, Singer-Lahat D, Freichel M et al. (1992) Calcium channel β subunit heterogeneity: functional expression of cloned cDNA from heart, aorta and brain. EMBO J 11:885–890
Beech DJ, Bernheim L, Mathie A, Hille B (1991) Intracellular Ca2+ buffers disrupt muscarinic suppression of Ca2+-current and M-current ion rat sympathetic neurons. Proc Natl Acad Sci USA 88:652–656
Bernheim L, Beech DJ, Hille B (1991) A diffusible second messenger mediates one of the pathways coupling receptors to calcium channels in rat sympathetic neurons. Neuron 6:859–867
Beech DJ, Bernheim L, Hille B (1992) Pertussis toxin and voltage dependence distinguish multiple pathways modulating calcium channels of rat sympathetic neurons. Neuron 8:97–106
Tsien RW, Tsien RY (1990) Calcium channels, stores, and oscillations. Annu Rev Cell Biol 6:715–760
Lipscombe D, Kongsamut S, Tsien RW (1989) Alpha-adrenergic inhibition of sympathetic neurotransmitter release mediated by modulation of N-type calcium-channel gating. Nature 340:639–642
Bean B (1989) Neurotransmitter inhibition of neuronal calcium currents by changes in channel voltage dependence. Nature 340:153–156
Delcour AH, Lipscombe D, Tsien RW (1992) Three modes of N-type calcium channel activity distinguished by differences in gating kinetics. J Neurosci (in press)
Flockerzi V, Oeken H-J, Hofmann F, Pelzer D, Cavalie A, Trautwein W (1986) Purified dihydropyridine-binding site from skeletal muscle T-tubules is a functional calcium channel. Nature 323:66–68
Nunoki K, Florio V, Catterall WA (1989) Activation of purified calcium channels by stoichiometric protein phosphorylation. Proc Natl Acad Sci USA 86:6816–6820
De Jongh KS, Warner C, Colvin AA, Catterall WA (1991) Characterisation of the two size forms of the α1 subunit of skeletal muscle L-type calcium channels. Proc Natl Acad Sci USA 88:10778–10782
Röhrkasten A, Meyer HE, Nastainczyk W, Sieber M, Hofmann F (1988) cAMP-dependent protein kinase rapidly phosphorylates serine-687 of the skeletal muscle receptor for calcium channel blockers. J Biol Chem 263:15325–15329
Rotman EI, De Jongh KS, Florio V, Lai Y, Catterall WA (1992) Specific phosphorylation of a C-terminal site on the full length form of the α1 subunit of the skeletal muscle calcium channel by cAMP-dependent protein kinase. J Biol Chem 267:16100–16105
Einhorn LC, Gregerson KA, Oxford GS (1991) D2 dopamine receptor activation of potassium channels in identified rat lactotrophs: whole-cell and single channel recording. J Neurosci 11:3727–3737
Smith PF, Luque EH, Neill JD (1986) Detection and measurement of secretion from individual neuroendocrine cells using a reverse hemolytic assay. Meth Enzymol 124:443–446
Lledo PM, Legendre P, Zhang J, Israel JM, Vincent JD (1990) Effects of dopamine on voltage-dependent potassium currents in identified rat lactotroph cells. Neuroendocrinology 52:545–555
Lledo PM, Legendre P, Israel JM, Vincent JD (1990) Dopamine inhibits two characterised voltage-dependent calcium currents in identified rat lactotroph cells. Endocrinology 127:990–1001
Stack J, Surprenant A (1991) Dopamine actions on calcium currents, potassium currents and hormone release in rat melanotrophs. I Physiol 439:37–58
Tse A, Hille B (1992) GnRH-induced Ca2+ oscillations and rhythmic hyperpolarisations of pituitary gonadotropes. Science 255:462–464
Marty A (1991) Calcium release and internal calcium regulation in acinar cells of exocrine glands. J Membr Biol 124:189–197
Kasai H, Augustine GJ (1990) Cytosolic Ca2+ gradients triggering unidirectional fluid secretion from exocrine pancreas. Nature 348:735–738
Marty A, Tan YP, Trautmann A (1984) Three types of Ca-dependent channels in rat lacrimal glands. J Physiol 357:293–325
Cook DL, Satin LS, Hopkins WF (1991) Pancreatic β-cells are bursting, but how? Trends Neurosci 14:411–414
Ashcroft SJH, Ashcroft FM (1989) The role of the ATP-sensitive K-channel in stimulus-response coupling in the pancreatic β-cell. In: Nunez J, Dumont JE (eds) Hormones and cell regulation, No 14. Colloque INSERM vol 198. J Libbey Eurotext, London Paris, pp 99–103
Bernardi H, Lazdunski M (1989) Pharmacology and regulation of ATP-sensitive K+ channels. Pflhgers Arch 414 [Suppl 1]:S80-S87
Niki I, Ashcroft SJH (1991) Possible involvement of protein phosphorylation in the regulation of the sulphonylurea receptor of a pancreatic beta-cell line, HIT T15. Biochem Biophys Acta 1133:95–101
Hopkins WF, Fatherazi S, Peter-Riesch B, Corkey BE, Cook DL (1992) Two sites for adenine-nucleotide regulation of ATP-sensitive potassium channels in mouse pancreatic β-cells and HIT cells. J Memb Biol (in press)
de Weille JR, Muller M, Lazdunski M (1992) Activation and inhibition of ATP-sensitive K+ channels by fluorescein derivatives. J Biol Chem 267:4557–4563
Niki I, Nicks JL, Ashcroft SJH (1990) The β-cell glibenclamide receptor is an ADP-binding protein. Biochem J 268:713–718
de Weille Jr, Schmid-Antomarchi H, Fosset M, Lazdunski M (1989) Regulation of ATP-sensitive K+ channels in insulinoma cells: activation by somatostatin and protein kinase C and the role of cAMP. Proc Natl Acad Sci USA 86:2971–2975
de Weille JR, Schmid-Antomarchi H, Fosset M, Lazdunski M (1988) ATP-sensitive K+ channels that are blocked by hypoic sulphonylureas in insulin secreting cells are activated by galanin, an hyperglycaemic hormone. Proc Natl Acad Sci USA 85:1312–1316
Amoroso S, Schmid-Antomarchi H, Fosset M, Lazdunski M (1990) Glucose, sulphonylureas, and neurotransmitter release: role of ATP-sensitive K+ channels. Science 247:852–854
Mourre C, Widmann C, Lazdunski M (1990) Sulfonylurea binding sites associated with ATP-regulated K+ channels in the central nervous system: autoradiographic analysis of their distribution and ontogenesis, and of their localisation in mutant mice cerebellum. Brain Res 519:29–43
Bernardi H, Fosset M, Lazdunski M (1988) Characterisation, purification, and affinity labelling of the brain [3H]glibenclami-debinding protein, a putative neuronal ATP-regulated K+ channel. Proc Natl Acad Sci USA 85:9816–9820
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Catterall, W., Epstein, P.N. Ion channels. Diabetologia 35 (Suppl 2), S23–S33 (1992). https://doi.org/10.1007/BF00586276
Issue Date:
DOI: https://doi.org/10.1007/BF00586276