Abstract
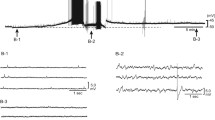
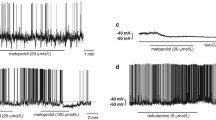
Postganglionic sympathetic neurons and brain noradrenergic neurons use α2A- and α2C-adrenoceptors as presynaptic autoreceptors. The present experiments were carried out in order to see whether they possess presynaptic α2B-autoreceptors as well. Pieces of atria, vasa deferentia, the occipito-parietal cortex and the hippocampus were prepared from either wildtype (WT) mice or mice in which both the α2A- and the α2C-adrenoceptor gene had been disrupted (α2ACKO). The pieces were incubated with 3H-noradrenaline and then superfused and stimulated electrically. In a first series of experiments, single pulses or brief, autoinhibition-poor pulse trains were used for stimulation. The α2-adrenoceptor agonist UK 14,304 (brimonidine) reduced the evoked overflow of tritium in all four tissues from WT mice but did not change it in any tissue from α2ACKO mice. A different pattern was obtained with medetomidine as α2 agonist. Like UK 14,304, medetomidine reduced the evoked overflow of tritium in all four tissues from WT mice and did not affect overflow in brain slices from α2ACKO mice; however, in contrast to UK 14,304, medetomidine reduced evoked overflow also in atrial and vas deferens pieces from α2ACKO mice, although with a lower maximum and potency than in WT preparations. The α-adrenoceptor antagonists rauwolscine, phentolamine, prazosin, spiroxatrine and WB 4101 shifted the concentration-response curve of medetomidine in α2ACKO atria and vasa deferentia to the right. The pKd values of the five antagonists against medetomidine in α2ACKO atria and vasa deferentia correlated with pKd values at prototypical α2B radioligand binding sites but not at α2A or α2C binding sites. In a second series of experiments, autoinhibition-rich pulse trains were used for stimulation. Under these conditions, rauwolscine and phentolamine increased the evoked overflow of tritium from α2ACKO atrial and vas deferens pieces but not from α2ACKO brain slices. The increase was smaller (by 40% in atria and by 70% in the vas deferens) than previously observed in WT preparations (by 200–400%). In a last series of experiments, mRNA for the α2B-adrenoceptor was demonstrated by RT-PCR in thoracolumbar sympathetic ganglia from WT, α2AKO, α2CKO and α2ACKO mice but not from α2BKO mice. The results show that brain noradrenergic neurons express only α2A- and α2C-adrenoceptors as autoreceptors. Postganglionic sympathetic neurons, however, can express α2B-adrenoceptors as presynaptic autoreceptors as well. The α2B-autoreceptors are activated by medetomidine but not by UK 14,304. They are also activated by previously released noradrenaline. The two-α2-autoreceptor hypothesis has to be replaced by a three-autoreceptor hypothesis for postganglionic sympathetic neurons.










Similar content being viewed by others
References
Altman JD, Trendelenburg AU, MacMillan L, Bernstein D, Limbird L, Starke K, Kobilka BK, Hein L (1999) Abnormal regulation of the sympathetic nervous system in α2A-adrenergic receptor knockout mice. Mol Pharmacol 56:154–161
Blaxall HS, Murphy TJ, Baker JC, Ray C, Bylund DB (1991) Characterization of the alpha-2C adrenergic receptor subtype in the opossum kidney and in the OK cell line. J Pharmacol Exp Ther 259:323–329
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159
Cox SL, Schelb V, Trendelenburg AU, Starke K (2000) Enhancement of noradrenaline release by angiotensin II and bradykinin in mouse atria: evidence for cross-talk between Gq/11 protein- and Gi/o protein-coupled receptors. Br J Pharmacol 129:1095–1102
Docherty JR (1998) Subtypes of functional α1- and α2-adrenoceptors. Eur J Pharmacol 361:1–15
Gaiser EG, Trendelenburg AU, Starke K (1999) A search for presynaptic imidazoline receptors at rat and rabbit noradrenergic neurones in the absence of α2-autoinhibition. Naunyn-Schmiedebergs Arch Pharmacol 359:123–132
Hein L (2001) Transgenic models of α2-adrenergic receptor subtype function. Rev Physiol Biochem Pharmacol 142:161–185
Hein L, Altman JD, Kobilka BK (1999) Two functionally distinct α2-adrenergic receptors regulate sympathetic neurotransmission. Nature 402:181–184
Jasper JR, Lesnick JD, Chang LK, Yamanishi SS, Chang TK, Hsu SAO, Daunt DA, Bonhaus DW, Eglen RM (1998) Ligand efficacy and potency at recombinant α2 adrenergic receptors. Agonist-mediated [35S]GTPγS binding. Biochem Pharmacol 55:1035–1043
Langer SZ (1977) Presynaptic receptors and their role in the regulation of transmitter release. Br J Pharmacol 60:481–497
Limberger N, Trendelenburg AU, Starke K (1992) Pharmacological characterization of presynaptic α2-autoreceptors in rat submaxillary gland and heart atrium. Br J Pharmacol 107:246–255
Limberger N, Funk L, Trendelenburg AU, Starke K (1995) Subclassification of presynaptic α2-adrenoceptors: α2A-autoreceptors in rabbit atria and kidney. Naunyn-Schmiedebergs Arch Pharmacol 352:31–42
Philipp M, Brede M, Hadamek K, Gessler M, Lohse MJ, Hein L (2002) Placental α2-adrenoceptors control vascular development at the interface between mother and embryo. Nat Genet 31:311–315
Sawamura S, Kingery WS, Davies MF, Agashe GS, Clark JD, Kobilka BK, Hashimoto T, Maze M (2000) Antinociceptive action of nitrous oxide is mediated by stimulation of noradrenergic neurons in the brainstem and activation of α2B adrenoceptors. J Neurosci 20:9242–9251
Simonneaux V, Ebadi M, Bylund DB (1991) Identification and characterization of the α2D-adrenergic receptors in bovine pineal gland. Mol Pharmacol 40:235–241
Starke K (1977) Regulation of noradrenaline release by presynaptic receptor systems. Rev Physiol Biochem Pharmacol 77:1–124
Starke K (2001) Presynaptic autoreceptors in the third decade: focus on α2-adrenoceptors. J Neurochem 78:685–693
Trendelenburg AU, Sutej I, Wahl CA, Molderings GJ, Rump LC, Starke K (1997) A re-investigation of questionable subclassifications of presynaptic α2-autoreceptors: rat vena cava, rat atria, human kidney and guinea-pig urethra. Naunyn-Schmiedebergs Arch Pharmacol 356:721–737
Trendelenburg AU, Hein L, Gaiser EG, Starke K (1999) Occurrence, pharmacology and function of presynaptic α2-autoreceptors in α2A/D-adrenoceptor-deficient mice. Naunyn-Schmiedebergs Arch Pharmacol 360:540–551
Trendelenburg AU, Klebroff W, Hein L, Starke K (2001) A study of presynaptic α2-autoreceptors in α2A/D-, α2B- and α2C-adrenoceptor-deficient mice. Naunyn-Schmiedebergs Arch Pharmacol 364:117–130
Trendelenburg AU, Meyer A, Klebroff W, Guimarães S, Starke K (2003a) Crosstalk between presynaptic angiotensin receptors, bradykinin receptors and α2-autoreceptors in sympathetic neurons: a study in α2-adrenoceptor-deficient mice. Br J Pharmacol 138:1389–1402
Trendelenburg AU, Meyer A, Klebroff W, Hein L, Starke K (2003b) Do all three α2-adrenoceptor subtypes operate as autoreceptors? Naunyn-Schmiedebergs Arch Pharmacol 367:R27
Vidovic M, Hill CE (1997) Transient expression of α-1B adrenoceptor messenger ribonucleic acids in the rat superior cervical ganglion during postnatal development. Neuroscience 77:841–848
Westfall TC (1977) Local regulation of adrenergic neurotransmission. Physiol Rev 57:659–728
Acknowledgement
This study was supported by the Deutsche Forschungsgemeinschaft (SFBs 487 und 505).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trendelenburg, AU., Philipp, M., Meyer, A. et al. All three α2-adrenoceptor types serve as autoreceptors in postganglionic sympathetic neurons. Naunyn-Schmiedeberg's Arch Pharmacol 368, 504–512 (2003). https://doi.org/10.1007/s00210-003-0829-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-003-0829-x