Abstract
Rationale
Nicotine produces behavioural effects that are potentially related to its interaction with diverse nicotinic acetylcholine receptor populations. Evidence from gene deletion studies suggests that the interoceptive stimulus properties of nicotine are mediated by heteromeric high-affinity receptors containing α4β2 subunits. Mice lacking β2 subunits do not discriminate nicotine (Shoaib et al., Neuropharmacology, 42:530–539, 2002), and nicotine does not elicit dopamine release in these animals (Grady et al., J Neurochem, 76:258–268, 2001). The stimulus properties of nicotine can be detected in rats using a two-lever operant drug discrimination paradigm, allowing them to be classified pharmacologically using ligands with selectivity for receptors containing α4β2, α3β4 or α7 subunits.
Materials and methods
Rats trained to discriminate 0.4 mg/kg nicotine from vehicle were given the nicotinic receptor agonists, cytisine, varenicline, TC2559, ABT-594, A-85380 (all having high affinity but varying selectivity for α4β2-containing receptors), and WO 03/062224 and WO 01/60821A1 (selective for β4- and α7-containing receptors, respectively). In separate studies, WO 03/062224 was used as the training stimulus.
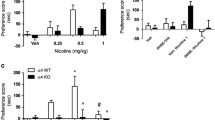
Results
Nicotine, TC-2559, A-85380 and ABT-594 showed dose-dependent and complete stimulus substitution, whilst WO 03/062224 and WO 01/60821A1 were completely without effect. Cytisine and varenicline showed partial generalisation, consistent with their partial agonist activity at nicotinic receptors eliciting dopamine release in rat striatal slices. After almost 50 training sessions with WO 03/062224, there was no clear evidence that an α3β4 receptor agonist could sustain a discriminable stimulus.
Conclusion
Substitution to the nicotine discriminative stimulus required high-affinity and high intrinsic activity at β2 but not at β4- or at α7-containing nicotinic receptors.







Similar content being viewed by others
References
Bannon AW, Decker MW, Kim DJB, Campbell JE, Arneric SP (1998) ABT-594, a novel cholinergic channel modulator, is efficacious in nerve ligation and diabetic neuropathy models of neuropathic pain. Brain Res 801:158–163
Brioni JD, Kim DJB, O’Neill AB (1996) Nicotine cue: lack of effect of the alpha7 nicotinic receptor antagonist methyllycaconitine. Eur J Pharmacol 301:1–5
Buckley MJ, Surowy C, Meyer M, Curzon P (2004) Mechanism of action of A-85380 in an animal model of depression. Prog Neuropsychopharmacol Biol Psychiatry 28:723–730
Cao YJ, Surowy CS, Puttfarcken PS (2005) Different nicotinic acetylcholine receptor subtypes mediating striatal and prefrontal cortical [3H]dopamine release. Neuropharmacology 48:72–79
Chandler CJ, Stolerman IP (1997) Discriminative stimulus properties of the nicotinic agonist cytisine. Psychopharmacology 129:257–264
Chen Y, Sharples TJW, Phillips KG, Benedetti G, Broad LM, Zwart R, Sher E (2003) The nicotinic alpha4beta2 receptor selective agonist, TC-2559, increases dopamine neuronal activity in the ventral tegmental area of rat midbrain slices. Neuropharmacology 45:334–344
Cheng YC, Prusoff WH (1973) Relationship between the inhibition constant (Ki) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol 22:3099–3108
Coe JW, Brooks PR, Wirtz MC, Bashore CG, Bianco KE, Vetelino MG, Arnold EP, Lebel LA, Fox CB, Tingley FD 3rd, Schulz DW, Davis TI, Sands SB, Mansbach RS, Rollema H, O’Neill BT (2005) 3,5-Bicyclic aryl piperidines: a novel class of alpha4beta2 neuronal nicotinic receptor partial agonists for smoking cessation. Bioorg Med Chem Lett 15:4889–4897
Cooper E, Couturier S, Ballivet M (1991) Pentameric structure and subunit stoichiometry of a neuronal acetylcholine receptor. Nature 350:235–238
Corrigall WA, Franklin KBJ, Coen KM, Clarke PBS (1992) The mesolimbic dopaminergic system is implicated in the reinforcing effects of nicotine. Psychopharmacology 107:285–289
Corrigall WA, Coen KM, Adamson KL (1994) Self-administered nicotine activates the mesolimbic dopamine system through the ventral tegmental area. Brain Res 653:278–284
Corringer PJ, Le Novere N, Changeux J-P (2000) Nicotinic receptors at the amino acid level. Annu Rev Pharmacol Toxicol 40:431–458
Couturier S, Bertrand D, Matter JM, Hernandez MC, Bertrand S, Millar N, Valera S, Barkas T, Ballivet M (1990) A neuronal nicotinic acetylcholine receptor subunit (alpha 7) is developmentally regulated and forms a homo-oligomeric channel blocked by alpha-BTX. Neuron 5:847–856
Cunningham CS, Polston JE, Jany JR, Segert IL, Miller DK (2006) Interaction of lobeline and nicotinic receptor ligands with the discriminative stimulus properties of cocaine and amphetamine. Drug Alcohol Depend 84:211–222
De Biasi M (2002) Nicotinic mechanisms in the autonomic control of organ systems. J Neurobiol 53:568–579
Di Chiara G (2000) Role of dopamine in the behavioural actions of nicotine related to addiction. Eur J Pharmacol 393:295–314
Di Chiara G, Imperato A (1988) Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc Natl Acad Sci USA 85:5274–5278
Donnelly-Roberts D, Puttfarcken PS, Kuntzweiler TA, Briggs CA, Anderson DJ, Campbell JE, Piattoni-Kaplan M, McKenna DG, Wasicak JT, Holladay MW, Williams M, Arneric SP (1998) ABT-594 [(R)-5-(2-azetidinylmethoxy)-2-chloropyridine]: a novel, orally effective analgesic acting via neuronal nicotinic acetylcholine receptors: I. In vitro characterisation. J Pharmacol Exp Ther 285:777–786
Fu Y, Matta SG, Sharp BM (1999) Local alpha-bungarotoxin-sensitive nicotinic receptors modulate hippocampal norepinephrine release by systemic nicotine. J Pharmacol Exp Ther 289:133–139
Garcha HS, Giardini V, Pratt JA, Stolerman IP (1983) Investigation of the effects of ganglion-blocking drugs and methergoline on the nicotine cue in rats. Br J Pharmacol 78:9P
Gommans J, Stolerman IP, Shoaib M (2000) Antagonism of the discriminative and aversive stimulus properties of nicotine in C57BL/6J mice. Neuropharmacology 39:2840–2847
Grady SR, Meinerz NM, Cao J, Reynolds AM, Picciotto MR, Changeux J-P, McIntosh JM, Marks MJ, Collins AC (2001) Nicotinic agonists stimulate acetylcholine release from mouse interpeduncular nucleus: a function mediated by a different nAChR than dopamine release from striatum. J Neurochem 76:258–268
Hirschhorn ID, Rosecrans JA (1974) Studies on the time course and the effect of cholinergic and adrenergic receptor blockers on the stimulus effect of nicotine. Psychopharmacologia 40:109–120
Kallman WM, Rosecrans JA, Kallman MJ, Harry GJ, Woodson PT (1982) Nicotine as a discriminative stimulus in human subjects. Psychopharmacology 76:A8
Kumar R, Reavill C, Stolerman IP (1987) Nicotine cue in rats: effects of central administration of ganglion-blocking drugs. Br J Pharmacol 90:239–246
Lambe EK, Picciotto MR, Aghajanian GK (2003) Nicotine induces glutamate release from thalamocortical terminals in prefrontal cortex. Neuropsychopharmacology 28:216–225
Le Novere N, Corringer P-J, Changeux J-P (2002) The diversity of subunit composition in nAChRs: evolutionary origins, physiologic and pharmacologic consequences. J Neurobiol 53:447–456
Leslie FM, Gallardo KA, Park MK (2002) Nicotinic acetylcholine receptor-mediated release of [3H]norepinephrine from developing and adult rat hippocampus: direct and indirect mechanisms. Neuropharmacology 42:653–661
Luo S, Kulak JM, Cartier GE, Jacobsen RB, Yoshikami D, Olivera BM, McIntosh JM (1998) alpha-conotoxin AuIB selectively blocks alpha3 beta4 nicotinic acetylcholine receptors and nicotine-evoked norepinephrine release. J Neurosci 18:8571–8579
Malin DH, Lake JR, Schopen CK, Kirk JW, Sailer EE, Lawless BA, Upchurch TP, Shenoi M, Rajan N (1997) Nicotine abstinence syndrome precipitated by central but not peripheral hexamethonium. Pharmacol Biochem Behav 58:695–699
Marks MJ, Pauly JR, Gross SD, Deneris ES, Hermans-Borgmeyer I, Heinemann SF, Collins AC (1992) Nicotine binding and nicotinic receptor subunit RNA after chronic nicotine treatment. J Neurosci 12:2765–2784
Miyata H, Ando K, Yanagita T (1999) Medial prefrontal cortex is involved in the discriminative stimulus effects of nicotine in rats. Psychopharmacology 145:234–236
Miyata H, Ando K, Yanagita T (2002) Brain regions mediating the discriminative stimulus effects of nicotine in rats. Ann N Y Acad Sci 965:354–363
Ohashi K, Osuga J-I, Tozawa R, Kitamine T, Yagyu H, Sekiya M, Tomita S, Okazaki H, Tamura Y, Yahagi N, Iizuka Y, Harada K, Gotoda T, Shimano H, Yamada N, Ishibashi S (2003) Early embryonic lethality caused by targeted disruption of the 3-hydroxy-3-methylglutaryl-CoA reductase gene. J Biol Chem 278:42936–42941
Picciotto MR, Zoli M, Rimondini R, Lena C, Marubio LM, Pich EM, Fuxe K, Changeux J-P (1998) Acetylcholine receptors containing the beta2 subunit are involved in the reinforcing properties of nicotine. Nature 391:173–177
Pratt JA, Stolerman IP, Garcha HS, Giardini V, Feyerabend C (1983) Discriminative stimulus properties of nicotine: further evidence for mediation at a cholinergic receptor. Psychopharmacology 81:54–60
Salas R, Orr-Urtreger A, Broide RS, Beaudet A, Paylor R, De Biasi M (2003) The nicotinic acetylcholine receptor subunit a5 mediates short-term effects of nicotine in vivo. Mol Pharmacol 63:1059–1066
Salas R, Cook KD, Bassetto L, De Biasi M (2004) The a3 and b4 nicotinic acetylcholine receptor subunits are necessary for nicotine-induced seizures and hypolocomotion. Neuropharmacology 47:401–407
Schechter MD (1995) Scopolamine–physostigmine combination does not substitute for nicotine. Prog Neuropsychopharmacol Biol Psychiatry 19:499–508
Shoaib M, Stolerman IP (1996) Brain sites mediating the discriminative stimulus effects of nicotine in rats. Behav Brain Res 78:183–188
Shoaib M, Zubaran C, Stolerman IP (2000) Antagonism of stimulus properties of nicotine by dihydro-b-erythroidine (DHbE) in rats. Psychopharmacology 149:140–146
Shoaib M, Gommans J, Morley A, Stolerman IP, Grailhe R, Changeux J (2002) The role of nicotinic receptor beta-2 subunits in nicotine discrimination and conditioned taste aversion. Neuropharmacology 42:530–539
Stolerman IP, Garcha HS, Pratt JA, Kumar R (1984) Role of training dose in discrimination of nicotine and related compounds by rats. Psychopharmacology 84:413–419
Stolerman IP, Chandler CJ, Garcha HS, Newton JM (1997) Selective antagonism of behavioural effects of nicotine by dihydro-beta-erythroidine in rats. Psychopharmacology 129:390–397
Stolerman IP, Chamberlain S, Bizarro L, Fernandes C, Schalkwyk L (2004) The role of nicotinic receptor alpha7 subunits in nicotine discrimination. Neuropharmacology 46:363–371
Sullivan JP, Donnelly-Roberts D, Briggs CA, Anderson DJ, Gopalakrishnan M, Piattoni-Kaplan M, Campbell JE, McKenna DG, Molinari E, Hettinger A-M, Garvey DS, Wasicak JT, Holladay MW, Williams M, Arneric SP (1998) A-85380 [3-(2(S)-azetidinylmethoxy) pyridine]: in vitro pharmacological properties of a novel, high affinity alpha4beta2 nicotinic acetylcholine receptor ligand. Neuropharmacology 35:725–734
Unwin N (2003) Structure and action of the nicotinic acetylcholine receptor explored by electron microscopy. FEBS Lett 555:91–95
Watkins SS, Epping-Jordan MP, Koob GF, Markou A (1999) Blockade of nicotine self-administration with nicotinic antagonists in rats. Pharmacol Biochem Behav 62:743–751
Wiley JL, James JR, Rosecrans JA (1996) Discriminative stimulus properties of nicotine: approaches to evaluating potential nicotinic receptor agonists and antagonists. Drug Dev Res 38:222–230
Wu J, George AA, Schroeder KM, Xu L, Marxer-Miller S, Lucero L, Lukas RJ (2004) Electrophysiological, pharmacological and molecular evidence for α7-nicotinic acetylcholine receptors in rat midbrain dopamine neurons. J Pharmacol Exp Ther 311:80–91
Xu W, Orr-Urtreger A, Nigro F, Gelber S, Sutcliffe CB, Armstrong D, Patrick JW, Role LW, Beaudet AL, De Biasi M (1999a) Multiorgan autonomic dysfunction in mice lacking the beta2 and the beta4 subunits of neuronal nicotinic acetylcholine receptors. J Neurosci 19:9298–9305
Xu W, Gelber S, Orr-Urtreger A, Armstrong D, Lewis RA, Ou C, Patrick J, Role L, De Biasi M, Beaudet AL (1999b) Megacystis, mydriasis, and ion channel defect in mice lacking the alpha3 neuronal nicotinic acetylcholine receptor. Proc Natl Acad Sci USA 96:5746–5751
Zhu PJ, Chiappinelli VA (2002) Nicotinic receptors mediate increased GABA release in brain through a tetrodotoxin-insensitive mechanism during prolonged exposure to nicotine. Neuroscience 115:137–144
Zoli M, Lena C, Picciotto MR, Changeux JP (1998) Identification of four classes of brain nicotinic receptors using beta2 mutant mice. J Neurosci 18:4461–4472
Acknowledgements
This work would not have been made possible without the technical assistance of Louise Wallace, Anne-Helene Clugery, Daniel Kumpik, Kimberley Smith, Ellen Colvin and John Boot.
All experiments conducted within this paper comply with the current laws of the country in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Smith, J.W., Mogg, A., Tafi, E. et al. Ligands selective for α4β2 but not α3β4 or α7 nicotinic receptors generalise to the nicotine discriminative stimulus in the rat. Psychopharmacology 190, 157–170 (2007). https://doi.org/10.1007/s00213-006-0596-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0596-8