Abstract
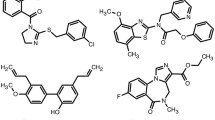
Phenol compounds, such as propofol and thymol, have been shown to act on the GABAA receptor through interaction with specific sites of this receptor. In addition, considering the high lipophilicity of phenols, it is possible that their pharmacological activity may also be the result of the interaction of phenol molecules with the surrounding lipid molecules, modulating the supramolecular organization of the receptor environment. Thus, in the present study, we study the pharmacological activity of some propofol- and thymol-related phenols on the native GABAA receptor using primary cultures of cortical neurons and investigate the effects of these compounds on the micro viscosity of artificial membranes by means of fluorescence anisotropy. The phenol compounds analyzed in this article are carvacrol, chlorothymol, and eugenol. All compounds were able to enhance the binding of [3H]flunitrazepam with EC50 values in the micromolar range and to increase the GABA-evoked Cl− influx in a concentration-dependent manner, both effects being inhibited by the competitive GABAA antagonist bicuculline. These results strongly suggest that the phenols studied are positive allosteric modulators of this receptor. Chlorothymol showed a bell-type effect, reducing its positive effect at concentrations >100 μM. The concentrations necessary to induce positive allosteric modulation of GABAA receptor were not cytotoxic. Although all compounds were able to decrease the micro viscosity of artificial membranes, chlorothymol displayed a larger effect which could explain its effects on [3H]flunitrazepam binding and on cell viability at high concentrations. Finally, it is suggested that these compounds may exert depressant activity on the central nervous system and potentiate the effects of general anesthetics.







Similar content being viewed by others
References
Macdonald, R. L., & Olsen, R. W. (1994). GABAA receptor channels. Annual Review of Neuroscience, 17, 569–602.
Rudolph, U., & Mohler, H. (2004). Analysis of GABAA receptor function and dissection of the pharmacology of benzodiazepines and general anesthetics through mouse genetics. Annual Review of Pharmacology and Toxicology, 44, 475–498.
Sogaard, R., Werge, T., Bertelsen, C., Lundbye, C., Madsen, K., Nielsen, C., et al. (2006). GABAA receptor function is regulated by lipid bilayer elasticity. Biochemistry, 45, 13118–13129.
Campagna-Slater, V., & Weaver, D. (2007). Anaesthetic binding sites for etomidate and propofol on a GABAA receptor model. Neuroscience Letters, 418, 28–33.
Mohammadi, B., Haeseler, G., Leuwer, M., Dengler, R., Krampfl, K., & Bufler, J. (2001). Structural requirements of phenol derivatives for direct activation of chloride currents via GABAA receptors. European Journal of Pharmacology, 421, 85–91.
García, D., Bujons, J., Vale, C., & Suñol, C. (2006). Allosteric positive interaction of thymol with the GABAA receptor in primary cultures of mouse cortical neurons. Neuropharmacology, 50, 25–35.
Johnston, G. (1996). GABAA receptor pharmacology. Pharmacology & Therapeutics, 69, 173–198.
Perillo, M., & García, D. (2001). Flunitrazepam induces geometrical changes at the lipid–water interface. Colloids and Surfaces B: Biointerfaces, 20, 63–72.
Sánchez, M. E., Turina, A. V., Garcia, D. A., Nolan, M. V., & Perillo, M. A. (2004). Surface activity of thymol: Implications for an eventual pharmacological activity. Colloids and Surfaces B: Biointerfaces, 34, 77–86.
Cantor, R. (2003). Receptor desensitization by neurotransmitters in membranes: Are neurotransmitters the endogenous anesthetics? Biochemistry, 42, 11891–11897.
Witt, M. R., & Nielsen, M. (1994). Characterization of the influence of unsaturated free fatty acids on brain GABA/benzodiazepine receptor binding in vitro. Journal of Neurochemistry, 62, 1432–1439.
García, D., Perillo, M., Zygadlo, J., & Martijena, I. (1995). The essential oil from Tagetes minuta L. modulates the binding of [3H]flunitrazepam to crude membranes from chick brain. Lipids, 30, 1105–1110.
Perillo, M., García, D., Marin, R., & Zygadlo, J. (1999). Tagetone modulates the coupling of flunitrazepam and GABA binding sites at GABAA receptor from chick brain membranes. Molecular Membrane Biology, 16, 189–194.
Pytel, M., Mercik, K., & Mozrzymas, J. (2006). Interaction between cyclodextrin and neuronal membrane results in modulation of GABAA receptor conformational transitions. British Journal of Pharmacology, 148, 413–422.
Reiner, G., Labuckas, D., & García, D. (2009). Lipophilicity of some GABAergic phenols and related compounds determined by HPLC and partition coefficients in different systems. Journal of Pharmaceutical and Biomedical Analysis, 49, 686–691.
García, D., Vendrell, I., Galofre, M., & Suñol, C. (2008). GABA released from cultured cortical neurons influences the modulation of t-[(35)S]butylbicyclophosphorothionate binding at the GABAA receptor effects of thymol. European Journal of Pharmacology, 600, 26–31.
Delgado Marín, L., Sánchez-Borzone, M., & García, D. A. (2011). Comparative antioxidant properties of some GABAergic phenols and related compounds, determined for homogeneous and membrane systems. Medicinal Chemistry, 7, 317–324.
Suñol, C., & Garcia, D. (2011. GABAA receptor binding and ion channel function in primary neuronal cultures for neuropharmacology/neurotoxicity testing. In M. Aschner, C. Suñol, & A. Bal-Price (Eds.), Neuromethods 56: cell culture techiques (pp. 481–493). New York: Humana Press.
Frandsen, A., & Schousboe, A. (1990). Development of excitatory amino acid induced cytotoxicity in cultured neurons. International Journal of Developmental Neuroscience, 8, 209–216.
Mosmann, T. (1983). Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. Journal of Immunological Methods, 65, 55–63.
Bangham, A., Hill, M., & Miller, N. (1974). Methods in membrane biology. New York: Plenum Press.
Lakowicz, J. (1986). Principles of fluorescence spectroscopy. New York: Plenum Press.
Hawkinson, J., Acosta-Burruel, M., Yang, K., Hogenkamp, D., Chen, J., Lan, N., et al. (1998). Substituted 3beta-phenylethynyl derivatives of 3alpha-hydroxy-5alpha-pregnan-20-one: Remarkably potent neuroactive steroid modulators of gamma-aminobutyric acidA receptors. Journal of Pharmacology and Experimental Therapeutics, 287, 198–207.
Suñol, C., Garcia, D. A., Bujons, J., Kristofikova, Z., Matyas, L., Babot, Z., et al. (2006). Activity of B-nor analogues of neurosteroids on the GABAA receptor in primary neuronal cultures. Journal of Medicinal Chemistry, 49, 3225–3234.
Fonfria, E., Rodriguez-Farre, E., & Suñol, C. (2001). Mercury interaction with the GABAA receptor modulates the benzodiazepine binding site in primary cultures of mouse cerebellar granule cells. Neuropharmacology, 41, 819–833.
Biltonen, R., & Lichtenberg, D. (1993). The use of differential scanning calorimetry as a tool to characterize liposome preparations. Chemistry and Physics of Lipids, 64, 129–142.
Sancar, F., & Czajkowski, C. (2011). Allosteric modulators induce distinct movements at the GABA-binding site interface of the GABAA receptor. Neuropharmacology, 60, 520–528.
Sigel, E., & Luscher, B. (2011). A closer look at the high affinity benzodiazepine binding site on GABAA receptors. Current Topics in Medicinal Chemistry, 11, 241–246.
Vale, C., Pomes, A., Rodriguez-Farre, E., & Suñol, C. (1997). Allosteric interactions between gamma-aminobutyric acid, benzodiazepine and picrotoxinin binding sites in primary cultures of cerebellar granule cells. Differential effects induced by gamma- and delta-hexachlorocyclohexane. European Journal of Pharmacology, 319, 343–353.
Vale, C., Vilaro, M. T., Rodriguez-Farre, E., & Suñol, C. (1999). Effects of the conformationally restricted GABA analogues, cis- and trans-4-aminocrotonic acid, on GABA neurotransmission in primary neuronal cultures. Journal of Neuroscience Research, 57, 95–105.
Bali, M., & Akabas, M. (2004). Defining the propofol binding site location on the GABAA receptor. Molecular Pharmacology, 65, 68–76.
De Vincenzi, M., Stammati, A., De Vincenzi, A., & Silano, M. (2004). Constituents of aromatic plants: Carvacrol. Fitoterapia, 75, 801–804.
Ipek, E., Tuylu, B., & Zeytinoglu, H. (2003). Effects of carvacrol on sister chromatid exchanges in human lymphocyte cultures. Cytotechnology, 43, 145–148.
Baser, K. (2008). Biological and pharmacological activities of carvacrol and carvacrol bearing essential oils. Current Pharmaceutical Design, 14, 3106–3119.
Jukic, M., Politeo, O., Maksimovic, M., & Milos, M. (2007). In vitro acetylcholinesterase inhibitory properties of thymol, carvacrol and their derivatives thymoquinone and thymohydroquinone. Phytotherapy Research, 21, 259–261.
Melo, F., Venancio, E., de Sousa, D., de Franca Fonteles, M., de Vasconcelos, S., Viana, G., et al. (2010). Anxiolytic-like effect of carvacrol (5-isopropyl-2-methylphenol) in mice: Involvement with GABAergic transmission. Fundamental & Clinical Pharmacology, 24, 437–443.
Kim, S., Oh, O., Min, H., Park, E., Kim, Y., Park, H., et al. (2003). Eugenol suppresses cyclooxygenase-2 expression in lipopolysaccharide-stimulated mouse macrophage RAW264.7 cells. Life Sciences, 73, 337–348.
Ohkubo, T., & Shibata, M. (1997). The selective capsaicin antagonist capsazepine abolishes the antinociceptive action of eugenol and guaiacol. Journal of Dental Research, 76, 848–851.
Cho, J., Kim, T., Lim, J., & Song, J. (2008). Effects of eugenol on Na+ currents in rat dorsal root ganglion neurons. Brain Research, 1243, 53–62.
Irie, Y., & Keung, W. (2003). Rhizoma acori graminei and its active principles protect PC-12 cells from the toxic effect of amyloid-beta peptide. Brain Research, 963, 282–289.
Wie, M. B., Won, M. H., Lee, K. H., Shin, J. H., Lee, J. C., Suh, H. W., et al. (1997). Eugenol protects neuronal cells from excitotoxic and oxidative injury in primary cortical cultures. Neuroscience Letters, 225, 93–96.
Won, M. H., Lee, J. C., Kim, Y. H., Song, D. K., Suh, H. W., Oh, Y. S., et al. (1998). Postischemic hypothermia induced by eugenol protects hippocampal neurons from global ischemia in gerbils. Neuroscience Letters, 254, 101–104.
Guenette, S., Beaudry, F., Marier, J., & Vachon, P. (2006). Pharmacokinetics and anesthetic activity of eugenol in male Sprague-Dawley rats. Journal of Veterinary Pharmacology and Therapeutics, 29, 265–270.
Müller, M., Pape, H. C., Speckmann, E. J., & Gorji, A. (2006). Effect of eugenol on spreading depression and epileptiform discharges in rat neocortical and hippocampal tissues. Neuroscience, 140, 743–751.
Aoshima, H., & Hamamoto, K. (1999). Potentiation of GABAA receptors expressed in Xenopus oocytes by perfume and phytoncid. Bioscience, Biotechnology, and Biochemistry, 63, 743–748.
Ultee, A., Kets, E. P., & Smid, E. J. (1999). Mechanisms of action of carvacrol on the food-borne pathogen Bacillus cereus. Applied and Environment Microbiology, 65, 4606–4610.
Gill, A., & Holley, R. (2006). Inhibition of membrane bound ATPases of Escherichia coli and Listeria monocytogenes by plant oil aromatics. International Journal of Food Microbiology, 111, 170–174.
Gill, A., & Holley, R. (2006). Disruption of Escherichia coli, Listeria monocytogenes and Lactobacillus sakei cellular membranes by plant oil aromatics. International Journal of Food Microbiology, 108, 1–9.
Wong, F. W., Lin, L., & Lim, D. C. (2009). Drug and herb interactions: Searching the web. Australian Family Physician, 38, 627–633.
Ang-Lee, M., Moss, J., & Yuan, C. (2001). Herbal medicines and perioperative care. JAMA, 286, 208–216.
Carrasco, M., Vallejo, J., Pardo-de-Santayana, M., Peral, D., Martin, M., & Altimiras, J. (2009). Interactions of Valeriana officinalis L. and Passiflora incarnata L. in a patient treated with lorazepam. Phytotherapy Research, 23, 1795–1796.
Vasiliadis, H. M., & Tempier, R. (2011). Reporting on the prevalence of drug and alternative health product use for mental health reasons: Results from a national population survey. Journal of Population Therapeutics and Clinical Pharmacology, 18, e33–e43.
Chen, X., Serag, E., Sneed, K., Liang, J., Chew, H., Pan, S., et al. (2011). Clinical herbal interactions with conventional drugs: From molecules to maladies. Current Medicinal Chemistry, 18, 4836–4850.
Cupp, M. (2000). Toxicology and clinical pharmacology of herbal products. Totowa, NJ: Humana Press.
Fugh-Berman, A. (2000). Herb-drug interactions. Lancet, 355, 134–138.
Kennedy, D., & Seely, D. (2010). Clinically based evidence of drug-herb interactions: A systematic review. Expert Opinion on Drug Safety, 9, 79–124.
Polzin, G. M., Stanfill, S. B., Brown, C. R., Ashley, D. L., & Watson, C. H. (2007). Determination of eugenol, anethole, and coumarin in the mainstream cigarette smoke of Indonesian clove cigarettes. Food and Chemical Toxicology, 45, 1948–1953.
Stanfill, S. B., Brown, C. R., Yan, X. J., Watson, C. H., & Ashley, D. L. (2006). Quantification of flavor-related compounds in the unburned contents of bidi and clove cigarettes. Journal of Agriculture and Food Chemistry, 54, 8580–8588.
Carlson, B. X., Engblom, A. C., Kristiansen, U., Schousboe, A., & Olsen, R. W. (2000). A single glycine residue at the entrance to the first membrane-spanning domain of the gamma-aminobutyric acid type A receptor beta(2) subunit affects allosteric sensitivity to GABA and anesthetics. Molecular Pharmacology, 57, 474–484.
Prete, P. S., Domingues, C. C., Meirelles, N. C., Malheiros, S. V., Goni, F. M., de Paula, E., et al. (2011). Multiple stages of detergent–erythrocyte membrane interaction—A spin label study. Biochimica et Biophysica Acta, 1808, 164–170.
Reiner, G. N., Perillo, M. A., & Garcia, D. A. (2013). Effects of propofol and other GABAergic phenols on membrane molecular organization. Colloids and Surfaces B: Biointerfaces, 101, 61–67.
Prendergast, F. G., Haugland, R. P., & Callahan, P. J. (1981). 1-[4-(Trimethylamino)phenyl]-6-phenylhexa-1,3,5-triene: Synthesis, fluorescence properties, and use as a fluorescence probe of lipid bilayers. Biochemistry, 20, 7333–7338.
Leonenko, Z. V., Finot, E., Ma, H., Dahms, T. E., & Cramb, D. T. (2004). Investigation of temperature-induced phase transitions in DOPC and DPPC phospholipid bilayers using temperature-controlled scanning force microscopy. Biophysical Journal, 86, 3783–3793.
Witzke, S., Duelund, L., Kongsted, J., Petersen, M., Mouritsen, O. G., & Khandelia, H. (2010). Inclusion of terpenoid plant extracts in lipid bilayers investigated by molecular dynamics simulations. The Journal of Physical Chemistry B, 114, 15825–15831.
Guenette, S., Helie, P., Beaudry, F., & Vachon, P. (2007). Eugenol for anesthesia of African clawed frogs (Xenopus laevis). Veterinary Anaesthesia and Analgesia, 34, 164–170.
Acknowledgments
This study was supported by Argentinian Grants FONCyT, CONICET, SECyT-UNC, Mincyt-Cba, IBRO, and by Spanish Grants from the Ministry of Science and Innovation (FIS 10/0453) and from the Generalitat de Catalunya (2009/SGR/214). D.A.G. and M.S.B. are members of CONICET (Argentina), G.N.R is a fellowship holder from the same institution, and L.D.M. is a fellowship holder from SECyT-UNC. N.O. is recipient of a CSIC contract in the JAE-Doc program cofinanced with European Social Funds.
Author information
Authors and Affiliations
Corresponding author
Additional information
G. N. Reiner and L. Delgado-Marín contributed equally to this study.
Rights and permissions
About this article
Cite this article
Reiner, G.N., Delgado-Marín, L., Olguín, N. et al. Gabaergic Pharmacological Activity of Propofol Related Compounds as Possible Enhancers of General Anesthetics and Interaction with Membranes. Cell Biochem Biophys 67, 515–525 (2013). https://doi.org/10.1007/s12013-013-9537-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-013-9537-4