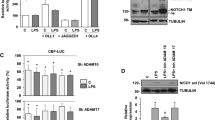
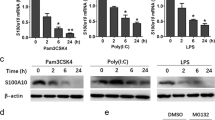
Abstract
Mammalian Toll-like receptors (TLRs) are expressed on innate immune cells and respond to the membrane components of Gram-positive or Gram-negative bacteria. When activated, they convey signals to transcription factors that orchestrate the inflammatory response. However, the intracellular signaling events following TLR activation are largely unknown. Here we show that TLR2 stimulation by Staphylococcus aureus induces a fast and transient activation of the Rho GTPases Rac1 and Cdc42 in the human monocytic cell line THP-1 and in 293 cells expressing TLR2. Dominant-negative Rac1N17, but not dominant-negative Cdc42N17, block nuclear factor-κB (NF-κB) transactivation. S. aureus stimulation causes the recruitment of active Rac1 and phosphatidylinositol-3 kinase (PI3K) to the TLR2 cytosolic domain. Tyrosine phosphorylation of TLR2 is required for assembly of a multiprotein complex that is necessary for subsequent NF-κB transcriptional activity. A signaling cascade composed of Rac1, PI3K and Akt targets nuclear p65 transactivation independently of IκBα degradation. Thus Rac1 controls a second, IκB–independent, pathway to NF-κB activation and is essential in innate immune cell signaling via TLR2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Aderem, A. & Ulevitch, R. J. Toll-like receptors in the induction of the innate immune response. Nature 406, 782–787 (2000).
Kopp, E. B. & Medzhitov, R. The Toll-receptor family and control of innate immunity. Curr. Op. Immunol. 11, 13–18 (1999).
Medzhitov, R., Preston-Hurlburt, P. & Janeway, C. A. A human homologue of the Drosophilia Toll protein signals activation of adaptive immunity. Nature 388, 394–397 (1997).
Lemaitre, B., Reichhart, J. M. & Hoffmann, J. A. Drosophila host defense: differential induction of antimicrobial peptide genes after induction by various classes of microorganisms. Proc. Natl Acad. Sci. USA 94, 14614–14619 (1997).
Rock, F. L., Hardiman, G., Timans, J. C., Kastelein, R. A., & Bazan, J. F. A family of human receptors structurally related to Drosophila Toll. Proc. Natl Acad. Sci. USA 95, 588–593 (1998).
Takeuchi, O. et al. Differential roles of TLR2 and TLR4 in recognition of Gram-negative and Gram-positive bacterial cell wall components. Immunity 11, 443–451 (1999).
Yoshimura, A. et al. Recognition of Gram-positive bacterial wall components by the innate immune system occurs via Toll-like receptor 2. J. Immunol. 163, 1–5 (1999).
Schwandner, R., Dziarski, R., Wesche, H., Rothe, M. & Kirschning, C. J. Peptidoglycan- and lipoteichoic acid-induced cell activation is mediated by Toll-like receptor 2. J. Biol. Chem. 274, 17406–17409 (1999).
Brightbill, H. D. et al. Host defense mechanisms triggered by microbial lipoproteins through toll-like receptors. Science 285, 732–736 (1999).
Kirschning, C. J., Wesche, H., Ayres, T. M. & Rothe, M. Human toll-like receptor 2 confers responsiveness to bacterial lipopolysaccharide. J. Exp. Med. 188, 2091–2097 (1998).
Yang, R. -B. et al. Toll-like receptor-2 mediates lipopolysaccharide-induced cellular signaling. Nature 395, 284–288 (1998).
Hirschfeld, M., Ma, Y., Weis, J. H., Vogel, S. N. & Weis, J. J. Repurification of lipopolysaccharide eliminates signaling through both human and murine Toll-like receptor 2. J. Immunol. 165, 618–622 (2000).
Bowie, A. & O'Neil, L. A. J. The interleukin-1 receptor/Toll-like receptor superfamily: signal generators for pro-inflammatory interleukins and microbial products. J. Leukoc. Biol. 67, 508–514 (2000).
Irie, T., Muta, T. & Takeshige, K. TAK1 mediates an activation signal from toll-like receptors to nuclear factor-κB in lipopolysaccharide-stimulated macrophages. FEBS Lett. 467, 160–164 (2000).
Mercurio, F. & Manning, A. M. Multiple signals converging on NF-κB. Curr. Op. Cell Biol. 11, 226–232 (1999).
Zandi, E. & Karin, M. Bridging the gap: composition, regulation, and physiological function of the IκB kinase complex. Mol. Cell. Biol. 19, 4547–4551 (1999).
Zhong, H., Sutang, H., Erdjument-Bromage, H., Tempst, P. & Ghosh, S. The transcriptional activity of NF-κB is regulated by the IκB-associated PKA subunit through a cyclic AMP-dependent mechanism. Cell 89, 413–424 (1997).
Van der Berghe, W. et al. p38 and extracellular signal-regulated kinase mitogen-activated protein kinase pathways are required for nuclear factor κB p65 transactivation mediated by tumor necrosis factor. J. Biol. Chem. 273, 3285–3290 (1998).
Sizemore, N., Leung S. & Stark, G. R. Activation of phosphatidylinositol 3-kinase in response to interleukin-1 leads to phosphorylation and activation of the NF-κB p65/RelA subunit. Mol. Cell. Biol. 19, 4798–4805 (1999).
Jefferies, C. A. & O'Neill, L. A. J. Rac1 regulates Interleukin 1-induced nuclear factor κB activation in an inhibitory protein κB-independent manner by enhancing the ability of the p65 subunit to transactivate gene expression. J. Biol. Chem. 275, 3114–3120 (2000).
Van Aelst, L. & D'Souza-Schorey, C. Rho GTPases and signaling networks. Genes Dev. 11, 2295–2322 (1997).
Chen, L. -M., Hobbie, S. & Galan, J. E. Requirement of Cdc42 for Salmonella-induced cytoskeletal and nuclear responses. Science 274, 2115–2118 (1996).
Lee, D. J., Cox, D., Li, J. & Greenberg, S. Rac1 and Cdc42 are required for phagocytosis but not NF-κB-dependent gene expression, in macrophages challenged with Pseudomonas aeruginosa. J. Biol. Chem. 275, 141–146 (2000).
Knaus, U. G., Heyworth, P. G., Evans, T., Curnutte, J. T. & Bokoch, G. M. Regulation of phagocyte oxygen radical production by the GTP-binding protein Rac 2. Science 254, 1512–1515 (1991).
Roberts, A. W. et al. Deficiency of the hematopoietic cell-specific Rho family GTPase Rac2 is characterized by abnormalities in neutrophil function and host defense. Immunity 10, 183–196 (1999).
Sulciner, D. J., Irani, K., Ferrans, V. J., Goldschmidt-Clermont, P. & Finkel, T. Rac1 regulates a cytokine-stimulated, redox-dependent pathway necessary for NF-κB activation. Mol. Cell. Biol. 16, 7115–7121 (1996).
Perona, R. et al. Activation of the nuclear factor-κB by Rho, Cdc42, and Rac1 proteins. Genes Dev. 11, 463–475 (1997).
Fruman, D. A. et al. Impaired B cell development and proliferation in absence of phosphoinositide 3-kinase p85α. Science 283, 393–397 (1999).
Benard, V., Bohl, B. J. & Bokoch, G. M. Characterization of Rac and Cdc42 activation in chemoattractant-stimulated human neutrophils using a novel assay for active GTPases. J. Biol. Chem. 274, 13198–13204 (1999).
Zenke, F., King, C. C., Bohl, B. P. & Bokoch, G. M. Identification of a central phosphorylation site in p21-activated kinase regulating autoinhibition and kinase activity. J. Biol. Chem. 274, 32565–32573 (1999).
Mira, J. -P., Benard, V., Groffen, J., Sanders, L. C. & Knaus, U. G. Endogenous hyperactive Rac3 controls proliferation of breast cancer cells by a p21-activated kinase-dependent pathway. Proc. Natl Acad. Sci. USA 97, 185–189 (2000).
Carpenter, C. L. & Cantley, L. C. Phosphoinositide kinases. Curr. Opin. Biol. 8, 153–157 (1996).
Zheng, Y., Bagrodia, S. & Cerione, R.A. Activation of phosphoinositide 3-kinase activity by Cdc42Hs binding to p85. J. Biol. Chem. 269, 18727–18730 (1994).
Bokoch, G. M., Vlahos, C. J., Wang, Y., Knaus, U. G. & Traynor-Kaplan, A. E. Rac GTPase interacts specifically with phosphatidylinositol 3-kinase. Biochem. J. 315,775–779 (1996).
Kandel, E. S. & Hay, N. The regulation and activities of the multifunctional Serine/Threonine kinase Akt/PKB. Exp. Cell Res. 253, 210–229 (1999).
Muzio, M., Natoli G., Saccani, S., Levrero, M. & Mantovani. The human toll signaling pathway: divergence of nuclear factor κB and JNK/SAPK activation upstream of tumor necrosis factor receptor-associated factor 6 (TRAF6). J. Exp. Med. 187, 2097–2101 (1998).
Montaner, S., Perona, R., Saniger, L. & Lacal, J. C. Multiple signaling pathways lead to the activation of the NF-κB by the Rho family of GTPases. J. Biol. Chem. 273, 12779–12785 (1998).
Singh, R. et al. The IL-1 receptor and Rho directly associate to drive cell activation in inflammation. J. Clin. Invest. 103, 1561–1570 (1999).
Puls, A. et al. Activation of the small GTPase Cdc42 by the inflammatory cytokines TNF and IL-1, and by the Epstein-Barr virus transforming protein LMP1. J. Cell Sci. 112, 2983–2992 (1999).
Vollebregt, M., Hampton, M. B. & Winterbourn, C. C. Activation of NF-κB in human neutrophils during phagocytosis of bacteria independently of oxidant generation. FEBS Lett. 432, 40–44 (1998).
Bird, T. A., Schooley, K., Dower, S. K., Hagen, H. & Virca, G. D. Activation of the transcription factor NF-κB by interleukin 1 is accompanied by casein kinase II-mediated phosphorylation of the p65 subunit. J. Biol. Chem. 272, 32606–32612 (1997).
Madrid, L.V. et al. Akt suppresses apoptosis by stimulating the transactivation potential of the RelA/p65 subunit of NF-κB. Mol. Cell. Biol. 20, 1626–1638 (2000).
Sahl, B., Wagey, R., Marotta, A., Tao, J. S. & Pelech, S. Activation of phosphatidylinositol 3-kinase, protein kinase B, and p70 S6 kinase in lipopolysaccharide-stimulated Raw 264.7 cells: differential effects of rapamycin, Ly294002, and wortmannin on nitric oxide production. J. Immunol. 161, 6947–6954 (1998).
Reddy, S. A. G., Huang, J. H. & Liao, W. S.-L. Phosphatidylinositol 3-kinase in interleukin 1 signaling. J. Biol. Chem. 272, 29167–29173 (1997).
Ozes, O. N. et al. NF-κB activation by tumor necrosis factor requires the Akt serine-threonine kinase. Nature 401, 82–86 (1999).
Romashkova, J. A. & Makarov, S.S. NF-κB is a target of Akt in anti-apoptotic PDGF signaling. Nature 401, 86–90 (1999).
Schiff, D. E. et al. Phagocytosis of Gram-negative bacteria by a unique CD14-dependent mechanism. J. Leuko. Biol. 62, 786–794 (1997).
Mercurio, F. et al. IKK-1 and IKK-2: cytokine-activated IkappaB kinases essential for NF-κB activation. Science 278, 860–866 (1997).
Sakurai, H., Chiba, H., Miyoshi, H., Sugita, T. & Toriumi, W. IκB kinases phosphorylate NF-κB p65 subunit on serine 536 in the transactivation domain. J. Biol. Chem. 274, 30353–30356 (1999).
Acknowledgements
We thank P. Rutledge for excellent secretarial support. Supported by NIH grants GM 37696 (to U. G. K. and to R. J. U.), GM 28485 and AI 15136 (to R. J. U.), HL48872 (to N.M.). N. T. is the recipient of a Boehringer Ingelheim Fonds fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arbibe, L., Mira, JP., Teusch, N. et al. Toll-like receptor 2–mediated NF-κB activation requires a Rac1-dependent pathway. Nat Immunol 1, 533–540 (2000). https://doi.org/10.1038/82797
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/82797
This article is cited by
-
Role of Rho GTPases in inflammatory bowel disease
Cell Death Discovery (2023)
-
Role of ELMO1 in inflammation and cancer—clinical implications
Cellular Oncology (2022)
-
Anfibatide Preserves Blood–Brain Barrier Integrity by Inhibiting TLR4/RhoA/ROCK Pathway After Cerebral Ischemia/Reperfusion Injury in Rat
Journal of Molecular Neuroscience (2020)
-
Amlexanox attenuates experimental autoimmune encephalomyelitis by inhibiting dendritic cell maturation and reprogramming effector and regulatory T cell responses
Journal of Neuroinflammation (2019)
-
Activation of Toll-like Receptor 2 (TLR2) induces Interleukin-6 trans-signaling
Scientific Reports (2019)